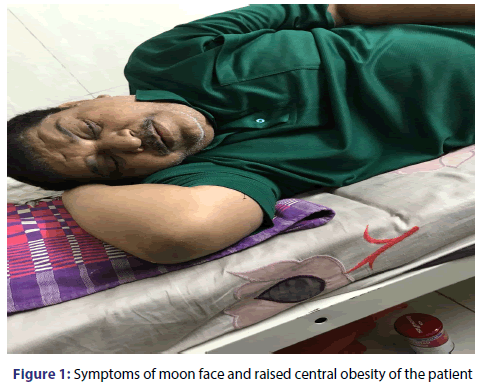
Prednisolone Induced Iatrogenic Cushing’s Syndrome Associated with Secondary Diabetes: A Case Report
Citation: Sateesh Kumar Reddy K, Ushasree P, Shanmuga Kumar SD, Goud SA. Prednisolone Induced Iatrogenic Cushing’s Syndrome Associated with Secondary Diabetes: A Case Report. J Basic Clin Pharma 2018;9:111-112.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@jbclinpharm.org
Abstract
Cushing’s syndrome is described as a complex conditions characterized by trunal hirsutism, purplish abdominal striae, edema, fragile skin and irregular menstruation for women. Occasionally, there may be changes in mood, headache, chronic feeling of tiredness and glucosuria. Cushing’s syndrome is an metabolic disorder due to the elevated glucocorticoid levels. It is broadly classified into two categories viz exogenous and endogenous. Many patients were affected by this disease due to the administration of exogenous glucocorticoids which are iatrogenic in nature. Here we report a case of 55 years old male patient claimed to have rapid weight gain after the treatment with prednisolone-15 mg/day for the rheumatoid arthritis for 15 months. He also been developing with peripheral symptoms such as moon face, central obesity, abdominal striae, edema over legs and symptoms of secondary diabetes mellitus. The patient has been recovered gradually by tapering the dose of Prednisolone and treated with hydroxychloroquine as a substituent for Rheumatoid arthritis.
Keywords
Cushing’s syndrome, prednisolone, secondary diabetes mellitus, central obesity
Introduction
Adverse drug reactions has been accounted over 6% of the total hospital admissions, this results in increase economic burden on health care system which is turn results in withdrawal of drugs from market.[1] Cushing syndrome is a systemic disorder caused by excessive activation of glucocorticoid receptors. It is most commonly iatrogenic, due to prolonged administration of synthetic glucocorticoids such as Prednisolone. Endogenous cushing’s syndrome is uncommon but is due to chronic over production of cortisol by the adrenal gland, either as the result of an adrenal tumor or because of excessive production of ACTH by a pituitary tumour or Actopic ACTH production by other tumor.[2] Cushing’s syndrome was discovered by the American neurosurgeon Harvey cushing in 1932. Cushing’s described a metabolic disorder characterized by truncal obesity, hypertension, fatigability and weakness, amenorrhea, diabetes mellitus, hirsutism, purplish abdominal striae, edema, glucosuria, osteoporosis and a basophilic tumor of the pituitary gland.[3] Cushing’s syndrome is four times more common in women than males.[4] The condition can be diagnosed by checking the previous medication treated for the patient for long duration and then measuring the cortisol in blood, urine and saliva after treating with dose of dexamethasone. The pathophysiology of cushing’s syndrome varies with the cause. In the treatment of iatrogenic Cushing’s syndrome the first step is to evaluate the exact cause of the disease and then either to reduce the dose of suspected drug or stop the treatment with that drug. Here we report an adverse drug reaction of Prednisolone which has been used to attenuate the symptoms of rheumatoid arthritis for the past 15 months.
A 55 years old male patient got admitted into the general medicine department of SVS medical college and hospital and presented with the complaints of moon face, rapid weight gain (76 kg to 115 kg) with in few months he also has complaints of abdominal striae, edema over legs, muscle weakness, fatigue, and the symptoms of secondary diabetes. Patient HBA1c levels were raised to 7.5 [Figure 1]. The past medical history shows he is suffering with rheumatoid arthritis for the last 15 months and which was diagnosed by anticyclic citrullinated peptide antibodies positive and rheumatoid factor assay shows 116.20 IU/ml, D- Dimer results were 1769.30 ng/ml, IgMRF (by ELISA) 271 IV/ml, all these findings provide evidence that patient has been suffering from rheumatoid arthritis, for which he had been treated with Prednisolone-15 mg/day, paracetamol-325 mg, serratiopeptidase-15 mg, and aceclofenac-100 mg BiD. The patient was oriented and the physical examination showed raised blood pressure to 130/90, pulse rate- 92 bpm, normal S1 and S2 sounds.
The laboratory investigations shows Glycosylated haemoglobin (HB1AC) 7.5, RBS-268 mg/dl, Hemoglobin-9 gm/dl, WBC-18,900 cells/cumm, platlet count-3.62 lakhs/cumm, total RBC count-4.34 mill/cumm, neutrophils-85%, total bilirubin- 0.50 mg/dl, total protein-6.30 gm/dl, alkaline phosphate-143 U/L, Albumin-3.10 gm/ dl, SGOT-21 U/L, SGPT-33 U/L, T3-0.915, T4-6.12 mcg/dl, TSH-2.75 Aμ IU/ml, serum potassium- 3.30 mmol/L, Serum cortisol levels were raised to 35 mcg/dl, failure to suppress cortisol with low dose of oral dexamethasone. 2D Echo cardiogram findings shows that LA-3.8 cm, A.O-3.4 cm, L.V: LVEDD-4.5 cm, EF-64%, LVESD-2.8 cm, F.S- 32%. From the subjective and objective evidence the patient was diagnosed from Cushing’s syndrome with secondary diabetes due to chronic use of oral prednisolone. Treatment started with tapering the dose of prednisolone to reduce complications such adrenal crisis. Patient has been treated with the following medication:- Tab. Prednisolone-5 mg OD, Tab. Hydroxychloroquine sulfate 200 mg/day, Inj. Ceftriaxone-1 g BD for 5 days; Tab. Metformin-500 mg BiD+Glimepiride-1 mg, Tab. Furosemide-20 mg/day, Tab. Ranitidine-150 mg/day, Tab. Paracetamol 325 mg+Aceclofenac 100 mg+Seretiopeptidase-15 mg BD. After 3 months the serum cortisol levels were measured and it shows reduced levels of serum cortisol.
Discussion
Cushing’s syndrome is caused due to the elevated glucocorticoid levels. Cushing’s syndrome can be broadly divided into exogenous and endogenous causes. The vast majority of cases of cushing’s syndrome are the results of the administration of exogenous glucocorticoids. The endogenous cause can inturn be divided into those that are ACTH dependent and those that are ACTH independent. Early stages of this disorder is manifested by hypertension, weight gain accompanied by central obesity, moon face, and accumulation fat in the posterior neck and back (buffalo hump). Glucocorticoids induce gluconeogensis and inhibit the uptake of glucose by cells with resultant hyperglycemia, glucosuria and polydipsia (secondary diabetes). The catabolic effects cause loss of collagen and reabsorption of bones. Skin becomes thin bruised, wound healing is poor and cutaneous striae are particularly common in the abdominal area, bone reabsorption results in the development of osteoporosis and increased susceptibility to fractures. Patients with cushing’s syndrome are at increased risk of variety of infections, because glucocorticoids suppress the immune response. Additional manifestations include several mental disturbances, including mood swings, depression and menstrual disturbances. [5] Patients with certain non-endocrine disorders may have some of the clinical or biochemical manifestations of cushing’s syndrome this may be confused with cushing’s syndrome, as well as 80% of patients with major depressive disorders have abnormally regulated cortisol secretion.[6] After confirming the serum cortisol levels by administering the low dose dexamethasone measurement of plasma ACTH is key to establishing the differential diagnosis. Low levels of ACTH or lack of response to ACTH stimulation test confirms the findings of cushing’s syndrome. Treatment is done by slowly tapering the dose of corticosteroid as the sudden stoppage may result in adrenal crisis. Slowly tapering the dose of steroid that is causing cushing’s syndrome can help reverse the effect of adrenal gland hypertrophy. [7] Furthermore the frequent use of multiple medication such as azole derivates (ketaconazole, itraconazole), macrolides and cisapride can put them at particular risk for Iatrogenic hypercorticism.[8] These drugs are metabolized by cytochrome P450 (CYP3A4), mostly present in the liver, and thereby inhibit metabolism of exogenous corticosteroids, since the latter are also partially metabolized by CYP3A4, and acts as inhibitors.[9] Resulting the Patient condition attenuated with the symptoms cushing’s syndrome. Serum cortisol levels reduced and the HBA1C levels reduced to 6.5% after 3 months of treatment. Patient should be educated about the adverse effects of corticosteroids when they were treated for long duration and the drug interactions such be properly monitored when the patient is undergoing treatment with multiple drugs to avoid complications.
REFERENCES
- Ratan JL, Lahkar M. A Study on drug induced Stevens Johnson Syndrome, toxic epidermal necrolysis, and SJS – TEN overlap in a tertiary care hospital of north east India. J Young Pharm 2016;8:149-53.
- Walker BR, Colledge NR, Ralston SH, Ian D. Davidsons principles and practice of medicine.pp: 773-6.
- Braunwald F, Hauser K, Jameson L. Harrison’s principles of internal medicine, pp: 2254-9.
- Sharma ST, Nieman L, Feelders RA. Cushing’s syndrome epidemiology and development indisease management. Clin Epidemiology 2015;7:281-93.
- Rabbins C. Pathologic basis of disease, pp: 1148-51.
- Ortho DN. Cushing’s syndrome. The New England Journal of Medicine 1995;332:791-803.
- Wisse B, Zieve D, Black B. Cushing syndrome exogenous. US national library of medicine. American Accreditation Health Care Commission.
- Wachter ED, Malfroot A, Schutter ID. Inhaled budesonide induced Cushing’s syndrome in the cystic fibrosis patients, due to drug inhibition of cytochrome P450. Journal of Cystic Fibrosis 2003;3:72-5.
- Vignes L, Archer VC, Diguet B. Effect of itraconazole on the pharmacokinetics of prednisolone and methylpresnisolone and cortisol secretion in healthy subjects. Br J Pharmacol 2001;51:443-50.