Evaluation of Anticancer Drug Utilization and Monitoring of Adverse Drug Reaction in the Indoor Patients Receiving Cancer Chemotherapy in a Tertiary Care Hospital in New Delhi
2 Department of Radiotherapy, Maulana Azad Medical College and Associated Hospitals, New Delhi, India
3 Department of Pharmacology, PGIMER and Dr RML Hospital, New Delhi, India
Citation: Aggarwal M, Chawla S, Singh K, Rana P. Evaluation of Anticancer Drug Utilization and Monitoring of Adverse Drug Reaction in the Indoor Patients Receiving Cancer Chemotherapy in a Tertiary Care Hospital in New Delhi. J Basic Clin Pharma 2018;9:118-122.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@jbclinpharm.org
Abstract
The main aim of drug utilization evaluation (DUE) is to promote the rational use of drugs in populations. The present study was designed to evaluate drug utilization pattern of anticancer drugs and to monitor the adverse drug reactions (ADR’s) in cancer patients receiving chemotherapy. Newly diagnosed patients of carcinoma which required treatment with chemotherapy, patients of both sex, and age>18 years were included in the study. Cisplatin (20% patients) is the most commonly prescribed anti-cancer drug in this study followed by 5-Fluorouracil (16% patients). Average number of drugs per prescription was 9.18 and percentage of drugs prescribed from Essential Drugs List (EDL) was 93.5%. ADR’s were observed in 93% of the study patients. The most commonly implicated group of anticancer agents in ADR’s, was platinum analog (32%) followed by antimetabolites (22%) and taxanes (20%). Antimicrobial agents use was observed to be restricted and judicious
Keywords
Carcinoma, chemotherapy, ADR, DUE, anticancer drugs
Introduction
Cancer is a group of diseases characterized by uncontrolled growth and spread of abnormal cells which can result in death if not controlled [1] it is a highly debilitating condition that is an important contributor to the global burden of disease. Worldwide, one in eight deaths is due to cancer.[1] Cancer causes more deaths than AIDS, tuberculosis and malaria combined. [1] It is the leading cause of death in developed countries and the second leading cause of death in developing countries (after cardiac disease).[1] An estimated 169.3 million years of healthy life were lost globally because of cancer in 2008.[2] By 2030, the global burden is expected to grow to approximately 21.4 million new cancer cases and 13.2 million cancer deaths, majority of which are in developing countries.[1] In India, noncommunicable diseases are the biggest cause of premature death.[3] The estimated number of new cancers in India is about 1.1 million per year.[4] More than 0.6 million people die of cancer each year[4] and approximately 42% cancers are tobacco related.[5] The main modalities used for cancer treatment include surgery, radiation, chemotherapy, hormones and immunotherapy. The choice of therapy depends upon patient factors, tumour factors and treatment factors.[6] Chemotherapy is the treatment of diseases with pharmacological agents.[7] Cancer chemotherapy utilizes anti-neoplastic drug or a combination of multiple drugs in a standardized regimen for the management of neoplasia.[8] It is the only therapy which acts systematically to eliminate disease from the whole body. [6] These drugs usually act on rapidly dividing cells and are either cell cycle specific or non-specific.[9]
The evaluation of drug utilization of anticancer drugs is imperative as their irrational use has created a major health problem in the current medical practice. Drug utilization has been defined by the WHO as the marketing, distribution, prescription and use of drugs in a society with special emphasis on the resulting medical and social consequences.[10] Drug utilization research facilitates the rational use of drugs in population.[10] Monitoring of drug utilization patterns helps to increase the therapeutic efficacy, provides feedback to the prescriber to ensure rational use of medicines and decrease the adverse drug reactions. The ultimate goal of drug utilization research must be to assess whether drug therapy is rational or not.[10] In spite all the cytotoxic benefits of cancer chemotherapy, they are common cause of morbidity as the drugs can harm healthy cells of the body. Cytotoxic agents do not differentiate between healthy and cancerous cells. All dividing cells exposed to these agents are killed resulting in significant adverse drug reactions (ADRs) in patients.[11]
ADRs occurring due to cancer chemotherapy significantly increase the cost of healthcare as well as an increase in morbidity and mortality. This leads to further increase in the suffering of the patient. Further, the ADR profile of cancer chemotherapeutics agents may differ in the Indian population due to genetic and ethnic differences.[12] The Adverse Drug Reactions recorded in patients in the present study can be a helpful tool in devising preventive measure for patients on cancer chemotherapy. The present study highlights the demographics of cancer patients, the commonly used cytotoxic drugs, the adjuvant palliative therapy given along with it and the adverse effects observed with anticancer drugs.
Materials and Methods
Drug utilization of anticancer drugs
Study design
The study was conducted as a prospective cross-sectional study.
Study site
The study was conducted in the cancer radiotherapy department of Maulana Azad Medical College and Lok Nayak hospital, New Delhi, one of the major tertiary care hospitals for cancer treatment, during a period from January 2013 to December 2013.
Inclusion criteria
Newly diagnosed patients of either sex, of any age and diagnosis admitted in Radiotherapy Department from January 2013 to August 2013 were included in the study. All the patients were followed for the complete duration of chemotherapy.
Exclusion criteria
Old cases i.e., patients already on chemotherapy.
Sample size
A total number of 101 patients were enrolled in the study from January 2013 to August 2013.
Ethical clearance
The study was approved by the Departmental Scientific Review Board and the Institutional Human Ethics Committee of Maulana Azad Medical College and Lok Nayak Hospital, New Delhi.
Adverse drug reaction monitoring of cancer patients
The ADR’s were recorded on the day of the 1st chemotherapy cycle and on all subsequent visits till the completion of the chemotherapy cycle. Information regarding the adverse drug reactions was collected using the standard form of the Central Drug Standard Control Organization (CDSCO). For the causality assessment Naranjo scale was used.
Observations and Results
Demographic profile
Age
The majority of patients (23.7%) were in the age group of 51-60 years followed by 19.8% in >60 years (20 patients), 18.81% in 41- 50 years (19 patients), 16.83% in 31-40 years (17 patients), 9.9% in 21-30 years (10 patients), 5.9% in 11-20 years (6 patients) and 4.9% in <10 years (5 patients). Approximately 62% patients were more than 40 years old. The mean age of the patients was 46.75 ± 18.25 years.
Sex
There were almost similar distribution of males and females in the study population. Females constituted 53% (54 patients) and males constituted 47% (47 patients) of the total population.
Education
Approximately 47.5% of the study population was illiterate (48 patients), 33.66% had attended primary school (34 patients), 10.89% attended up to secondary school (11 patients), 4.95% patients attended senior secondary school (5 patients) and 2.97% patients were graduates.
Patient diagnosis
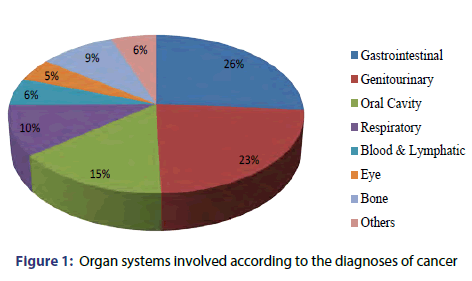
Carcinoma (Ca) of the oral cavity was the most common diagnosis (16% patients) followed by Ca cervix and Anorectal Ca (9% patients each), Ca oesophagus and Ca Ovary (8% patients each), Ca colon (6% patients), Ca larynx (5% patients), Ca breast and Ca lungs (4% patients each), Retinoblastoma and Ca urinary bladder (3% patients each). Round Cell Tumor Eye, Hodgkin’s Lymphoma, Osteosarcoma, Ca Gall Bladder, Ca Maxilla, Plasmacytoma, Basal Cell Carcinoma accounted for 2% patients each followed by other diagnosis (11% patients) [Figure 1].
Anticancer drug utilization pattern
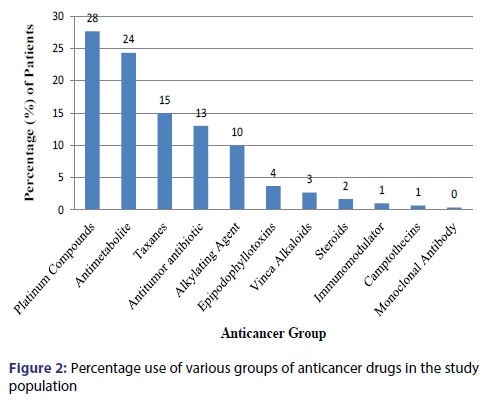
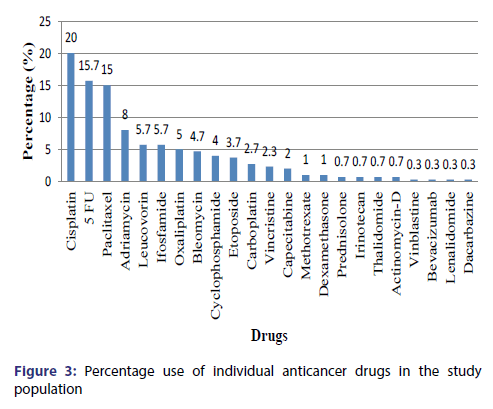
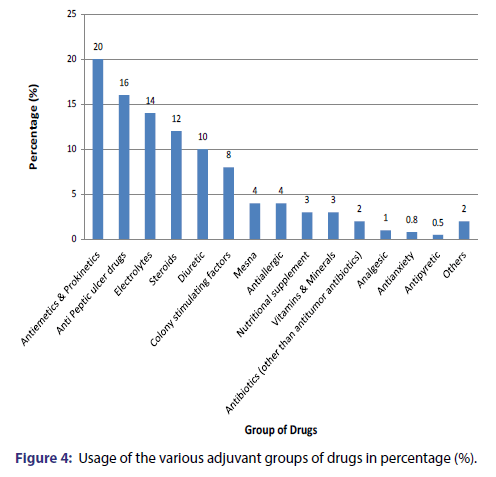
The most commonly used class of anticancer agents were the Platinum Compounds (28% patients) followed by Antimetabolites (24% patients), Taxanes (15% patients), Antitumor antibiotic (13% patients), Alkylating Agent (10.3% patients), Epipodophyllotoxins (3.7% patients) [Figure 2]. The most commonly used drug were Cisplatin (20% patients) followed by 5-Fluorouracil (16% patients), Paclitaxel (15% patients), Adriamycin (7.7% patients), Leucovorin and Ifosfamide (5.7% patients), Oxaliplatin(5% patients), Bleomycin(4.7% patients), Cyclophosphamide(4% patients), Etoposide(3.7% patients), Carboplatin(2.7% patients) [Figure 3]. Approximately 96% of the cytotoxic drugs were given in injectable form and 4% drugs were given orally. Majority (75%) of drugs were given at 3 weeks interval followed by a frequency of 2 weeks (16%), 4 weeks (7%) and 1 week (2%). Various adjuvant drugs like anti-emetics and anti-peptic ulcer drugs were also used along with anticancer agents [Figure 4].
Adverse drug reactions
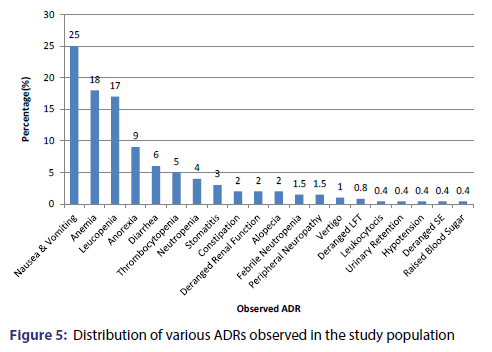
ADR’s were observed in 93% of the patients. About 48% of these patients were males and 52% patients were females. These patients most commonly belonged to the age group 50-60 years (26.8%). The second commonest age group showing adverse drug reaction was >60 years (18.29%) followed by the 40-50 years age group (17.07%) and 30-40 years (15.85%). Based on the Naranjo Adverse Drug Reaction probability scale, 1% of the adverse effects were certain, 73% were probable and 26% were possible. A total of 262 adverse effects were observed. There were of 20 different types. The most common adverse effect was nausea and vomiting (25%) followed by anaemia (18%) leukopenia (17%), anorexia (9%), diarrhoea (6%), thrombocytopenia (5%), neutropenia (4%) and stomatitis (3%) [Figure 5].
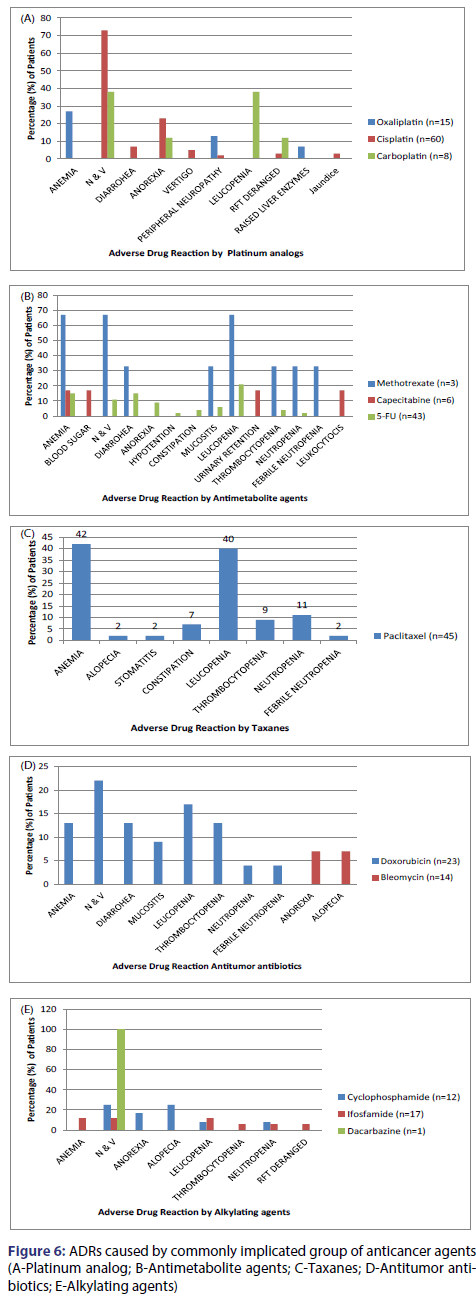
The most commonly implicated group of anticancer agents was platinum analog (32%) followed by antimetabolites (22%), taxanes (20%), antitumor antibiotics (9%), epipodophyllotoxin and alkylating agents (8% each) and camptothecins (1%) [Table 1]. The most common causal drug was Cisplatin (26.33%), followed by Paclitaxel (19.8%), 5-Fluorouracil (16.4%), Etoposide and Doxorubicin (8% each), Methotrexate and Cyclophosphamide (4% each), Ifosfamide (3.4% each), Carboplatin and Oxaliplatin (in 3% each), Capecitabine (1.5%), Irinotecan (1%), Bleomycin and Dacarbazine (<1% each) [Figure 6].
| ADRs | Platinum compounds % (n=83) |
Antimetabolite % (n=73) |
Taxanes % (n=45) |
Antitumor antibiotic % (n=39) |
Alkylating Agent % (n=30) |
Epipodophyllotoxins % (n=11) |
Camptothecins % (n=2) |
|---|---|---|---|---|---|---|---|
| Nausea and Vomiting | 57 (47) | 10 (7) | - | 13 (5) | 20 (6) | 9 (1) | - |
| Anorexia | 18 (15) | 5 (4) | - | 3 (1) | 7 (2) | 9 (1) | - |
| Diarrhea | 5 (4) | 11 (8) | - | 8 (3) | - | - | - |
| Stomatitis | - | 6 (4) | 2 (1) | 5 (2) | - | 18 (2) | - |
| Constipation | 3 (2) | 7 (3) | - | - | - | ||
| Anemia | 5 (4) | 12 (9) | 42 (19) | 8 (3) | 7 (2) | 73 (8) | - |
| Leucopenia | 4 (3) | 16 (12) | 40 (18) | 10 (4) | 10 (3) | 36 (4) | 50 (1) |
| Thrombocytopenia | - | 4 (3) | 9 (4) | 8 (3) | 3 (1) | 27 (3) | - |
| Neutropenia | - | 3 (2) | 11 (5) | 3 (1) | 7 (2) | 9 (1) | - |
| Febrile Neutropenia | - | 2 (1) | 2 (1) | 3 (1) | - | 9 (1) | - |
| Leukocytocis | - | 2 (1) | - | - | - | - | - |
| Urinary Retention | - | 2 (1) | - | - | - | - | - |
| Deranged Renal Function | 4 (3) | - | - | - | 3 (1) | - | 50 (1) |
| Peripheral Neuropathy | 4 (3) | 2 (1) | - | - | - | - | - |
| Vertigo | 4 (3) | - | - | - | - | - | - |
| Deranged LFT | 1 (1) | - | - | - | - | - | - |
| Alopecia | - | - | 2 (1) | 3 (1) | 10 (3) | - | - |
| Hypotension | - | 2 (1) | - | - | - | - | - |
| Deranged SE | - | - | - | - | - | - | 50 (1) |
| Raised Blood Sugar | - | 2 (1) | - | - | - | - | - |
Table 1: Group of anticancer drugs prescribed versus the episodes of ADR encountered.
Core drug use indicators
The WHO core drug use indicators were also evaluated in this study. The prescribing indicators and health facility indicators were studied and their results are summarised in Table 2.
| 2a. Prescribing indicators | ||
| Serial No. | Prescribing indicators | Value |
| 1. | Average number of drugs per encounter | 9.18 ± 2.74 |
| 2. | Percentage of drugs prescribed by generic name (%) | 69.9 |
| 3. | Percentage of encounters with an antibiotic prescribed (%) | 7.90 |
| 4. | Percentage of encounters with an injection prescribed (%) | 100 |
| 5. | Percentage of drugs prescribed from essential drugs list (%) | 93.5 |
| 6. | Percentage of anticancer drugs prescribed from essential drugs list (%) | 98 |
| 2b. Health facility indicators | ||
| Serial No. | Health Facility indicators | Value |
| 1 | Availability of copy of essential drugs list | Yes |
| 2 | Availability of anticancer drugs (% of anticancer drugs available in the essential drugs list) | 32.35 |
Table 2: WHO core drug use indicators
Discussion
The principal aim of a drug utilization study is to promote the rational use of drugs in populations. The present study shows that the most common class of cytotoxic agents prescribed was platinum compounds (28%). Amongst the platinum analogs, the most commonly used drug was cisplatin (72%) which was given to about 20% of all the patients. The other more commonly prescribed drugs were 5-fluorouracil (16%), paclitaxel (15%),s doxorubicin (8%), leucovorin (6%), ifosfamide (6%) and oxaliplatin (5%). The results are similar to the drug utilization study done in Nepal by Muhammad Khan et al.[13]
The most prevalent cancer in the study population was oral cavity cancer and the regimen used most commonly used for its treatment was the PCF regimen, consisting of cisplatin, paclitaxel and 5-Fluorouracil which also resulted in these 3 drugs being the most used cytotoxic agents in our study. The second commonest class used was antimetabolites (26%) amongst which most used was 5-fluorouracil (65%) followed by leucovorin (23%), capecitabine (8%) and methotrexate. 5-fluorouracil, leucovorin and oxaliplatin were used together as the FOLFOX-7 regimen for the treatment of colorectal cancer. Similarly, methotrexate was most commonly used as a part of palliative therapy in patients with advanced and incurable cancers.
After antimetabolites, anti-mitotic and antitumor antibiotic were the 3rd and 4th commonest class of agents used. The antimitotic plant derivatives consisted of taxanes (15%), epipodophyllotoxins (3.5%), vinca alkaloids (2.5%) and camptothecins (1%). Amongst the antitumor antibiotics which together accounted for 13% drug use, the commonest doxorubicin (65%) followed by bleomycin (35%) and actinomycin-D (5%). This was followed by alkylating agents, which included nitrogen mustards: cyclophosphamide (57%) was the commonest followed by ifosfamide (40%) and dacarbazine (3%). These findings are in tune with the drug utilization study done in Nepal by Muhammad Khan et al.[13] with alkylating agents (46%) as the most commonly used drug class. The antimetabolites (22%) were the second most common class of drugs given. Cytotoxic antibiotics and antimitotic plant derivatives were given in 14% patients each and hormonal agents were given in 4% of the patients.[13]
In our study, the frequency of administration of the anticancer drugs was noted. Majority of the drugs (94%) were given once a day. Continuous infusion over 48 hours was used in 5% patients for the administration of 5-fluorouracil in the FOLFOX regimen. The purpose for using this method for 5-FU administration was to decrease the incidence of diarrhea, which occurs as an adverse effect of its bolus administration. Capecitabine was prescribed to 6 patients, but it was the only cytotoxic drug to be given orally (in 2 patients). The frequency of the cancer chemotherapy cycle was 3 weeks in majority (75%) of the patients. The reason for this is that 3 weeks is usually considered the optimum amount of time to recover from the adverse effects of chemotherapy, especially bone marrow depression, an ADR caused by majority of the anticancer agents. The second commonest frequency (16%) was of 2 weeks, used especially in FOLFOX regimen.
Cancer chemotherapy includes cytotoxic medicines accompanied by adjuvant and supplementing therapeutic measures. These additional medications other than the cytotoxic medicine are for reducing the adverse effects seen with the cancer chemotherapy. The average number of other drugs in our study was 6.2 per prescription. On an average 75% of the patients were prescribed a steroid along with the chemotherapy. The adjuvant steroid used with chemotherapy in order to minimize the adverse effects which included nausea, vomiting and anorexia was dexamethasone.[14] It alleviates nausea caused by chemotherapy medications, stimulates appetite, decreases inflammation at the cancer site, and also decreases the elevated blood calcium levels (associated with some bone cancer cases).[15] All the patients were on an average prescribed either one vitamin or mineral substitute. Potassium chloride and Magnesium sulphate were mineral supplements given to almost all the patients receiving a cisplatin based chemotherapy regimen. Cisplatin is a common cause of hypomagnesaemia and hypokalemia due to renal magnesium (Mg) and potassium (K) losses. Magnesium plays an important role in the maintenance of intracellular K.[16] Prophylactic magnesium supplementation, in addition to preventing adverse effects that result directly from magnesium deficiency, can decrease the severity of cisplatin-induced renal damage without interfering with the anticancer effect of the drug.[17] Similarly, patients on Cisplatin have a high chance of renal tubular dysfunction and a cumulative impairment in renal function (as manifested by a decline in the glomerular filtration rate).[18] In order to prevent the development nephrotoxicity, forced hydration in the form of saline infusion on the day of chemotherapy, prior to and following cisplatin (total of 3.5- 4.0 liters during 3-4 hours)[19] and diuresis (by mannitol or furosemide) was given to the patient’s in our study.
Administration of cytotoxic drugs is associated in some patients with severe acid reflux diseases which warrant the administration of proton pump inhibitors, H2 antagonist and antacid prophylactically as well as therapeutically to the patients. [20] On an average, one anti peptic ulcer drug was given to each patient. Approximately 50% of the patients were given colony stimulating factor and one fourth patients were prescribed mesna. The other drugs prescribed included zoledronic acid, lactic acid, lactobacillus etc. Mesna is used along with cyclophosphamide and ifosfamide to prevent hemorrhagic cystitis and antiallergics are prescribed with cisplatin to prevent anaphylactic reaction.
There were only 7.9% encounters with an antimicrobial prescribed. On an average 0.10 antimicrobials were prescribed per prescription. These numbers are less than the findings of the study done in Nepal in which 31.25% encounters had an antibiotic prescribed with an average of 0.37 antibiotics per prescription.[16] This result is due to the fact that most of the antimicrobials in that study included the antitumor antibiotics that were prescribed according to the respective regimen of cancer type. But on analyses of non-antitumor antibiotics, they found that the occurrence of antibiotics per prescription was just 9.16%. This is an encouraging finding and suggests rational prescribing of antimicrobial agents. Our results are comparable with the drug utilization study done in Nepal by Muhammad Khan et al.[13] in which the average number of other prescribed per prescription was 1.67 antiemetic’s, 1.5 supplements (iron, vitamin and protein), 1.13 anti-peptic ulcer drug (including proton pump inhibitors and H2 antagonist) and 0.97 steroids. In our study, 70% of the drugs were prescribed using the generic name of the drug and approximately 93% of the prescribed drugs were from the essential medicine list. For the cytotoxic drugs, 96% were prescribed using generic name, which is an indicator of rational prescribing.
In our study, 93% patients had an ADR to the cancer chemotherapy. These observations were similar to a study done by Datta et al.[21] and De [22], in which 86% and 98% patients respectively developed ADR due to chemotherapy. The demographic profile of patients’ presenting with adverse drug was representative of the population, indicating that the patients’ receiving the drug had the highest probability of developing ADRs. Various adverse effects were observed involving eight organ systems. Chemotherapy kills rapidly dividing cells. Most severely affected organ systems included haematological and gastrointestinal system, each accounting for 46% of all ADRs. The most common adverse effect in the study population was nausea and vomiting observed in 65% of all the patients. This was attributed to platinum compounds in majority of the cases, amongst which cisplatin was the most emetogenic, accounting for 67% cases. Approximately 73% patients taking Cisplatin developed either nausea or vomiting. The most common mechanism of chemotherapy induced nausea and vomiting is through activation of Chemoreceptor Trigger Zone (CTZ). Studies done for monitoring ADRs to cancer chemotherapy found CINV (Chemotherapy-induced nausea and vomiting) as the commonest adverse effect following administration of chemotherapeutic agents.[21-26] The incidence of CINV varied from 48% [22] to 60% [25] in the study population.
Other GIT manifestations included anorexia, diarrhoea, and constipation and these were present in 23%, 15% and 5% patients respectively. In the present study, patients on antimetabolites had the highest incidence of diarrhoea especially 5-Fluorouracil, which causes this adverse effect when given as a bolus injection. To prevent this ADR, it is usually given as a 48 hour infusion to the patient. Bolus 5-FU causes leukopenia, mucositis, and diarrhoea whereas continuous infusion 5-FU causes more stomatitis.[27] Approximately 15% patients in our study developed diarrhoea. The incidence in other studies ranged from 1% [22] to 17%. [24] The second commonest adverse effect in our study was anaemia followed by leukopenia, seen in 46% and 45% of patients respectively. Other hematopoietic manifestations included thrombocytopenia (14%) and neutropenia (10%). In majority of the previous studies done to monitor ADR due to chemotherapy, bone marrow suppression was the second commonest adverse effect observed, with the incidence ranging from 14% to 35%. [21-26]
In the present study, patients on etoposide had the highest incidence of development of anaemia (73%) and thrombocytopenia (27%) followed by paclitaxel (42% and 9% respectively). Patients on paclitaxel had the highest incidence of leukopenia (40%) and neutropenia (11%) followed by etoposide (36% and 9% respectively). Other less common but highly debilitating adverse effects included nephrotoxicity (manifested as increase in serum urea/creatinine levels) and ototoxicity (vestibulotoxicity manifesting as vertigo), occurring in patients on cisplatin. Nephrotoxicity and ototoxicity were seen in 5% and 3% of the patients respectively. In our study, cisplatin was the drug causing maximum (27%) ADRs. This result was similar to the study done by Datta [21] and De, in which Cisplatin was responsible for 29% and 26% of the total ADRs respectively. The ADRs caused by it included anorexia, nausea and vomiting, vertigo, deranged kidney and liver function, diarrhoea and peripheral neuropathy. Cisplatin being the most frequently incriminated drug does not necessarily mean that it is the one most prone to cause ADRs; it may reflect the fact that cisplatin is one of the most widely used anticancer drugs in that unit.
The other commonly implicated drugs were paclitaxel (20%), 5 FU (16%) and doxorubicin (8%). Paclitaxel caused myelosuppression, constipation and alopecia. 5 FU affected the gastrointestinal tract (diarrhoea, stomatitis, anorexia, nausea and vomiting) in addition to causing myelosuppression, peripheral neuropathy and hypotension. The adverse effects caused by doxorubicin included nausea and vomiting, bone marrow suppression, diarrhoea and stomatitis. Causality assessment of ADRs was done using naranjo’s adverse drug reaction probability scale.[28] Naranjo scale is one of the commonest methods used in research for causality assessment of ADRs. Seventy three percent ADRs were found to be probable, 26% were possible and 1% was definite. The definite adverse effects included two cases of severe vomiting, caused by methotrexate and cisplatin and one case of mucositis in a patient on methotrexate alone. In a study done to assess the pattern of adverse drug reactions due to cancer chemotherapy in a tertiary care teaching hospital in Bhubaneswar, 52 adverse effects were observed, out of which 62% were found to be probable, 31% were possible and 7% were definite. Another study was done for monitoring of adverse drug reactions in the oncology unit of a multispecialty teaching hospital in Kolkata evaluated the 295 suspected adverse effects and found 85% adverse effects to be probable, 13% as possible and 2% as definite.
Conclusion
From the present study, we can conclude that cisplatin is the most commonly used cytotoxic drug followed by 5-FU and paclitaxal. ADR’s were observed in 95% of the study patients. The GIT manifestations like nausea and vomiting followed by diarrhea were the commonest ADRs reported, these were strongly attributed to the use of platinum compounds (cisplatin) and antimetabolites (5-FU) respectively. The most commonly used adjuvant drugs in our study were anti-emetics and antipeptic ulcer drugs. Antimicrobial agents use was observed to be restricted and judicious. Over 93% of cytotoxic agents were chosen from the essential drug list and were prescribed in generic names, indicating rational use. WHO advocates that more such drug utilization studies are needed in every health care setting to assess and assure the rational drug use.
REFERENCES
- American Cancer Society. Global Cancer Facts and Figures 2nd Edition 2011 Available from: http://www.cancer.org/research/cancerfactsfigures/globalcancerfactsfigures/global-facts-figures-2nd-ed
- Cancer research UK. Cancer worldwide-cancer statistics key facts 2014 Available from: http://publications.cancerresearchuk.org/downloads/Product/CS_KF_WORLDWIDE.pdf
- Haskell CM. Introduction. Cancer treatment 4th Edition, Philadelphia, WB Saunders 1995;3-9.
- American Cancer Society. Chemotherapy principles 2013. Available from: URL:http://www.cancer.org/treatment/treatmentsandsideeffects/treatmenttypes/chemotherapy/chemotherapyprinciplesanin-depthdiscussionofthetechniquesanditsroleintreatment/chemotherapy-principles-what-is-chemo
- Times of India. 48% cancers due to tobacco chewing 2011. Available from: https://timesofindia.indiatimes.com/city/chandigarh/48-cancers-due-to-tobacco-chewing/articleshow/10649252.cms?
- askell CM. Introduction. In: Haskell CM, editor. Cancer treatment 4th ed. Philadelphia, W.B. SAUNDERS;1995. p 3-9.
- American Cancer Society. Chemotherapy principles 2013. Available from: https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/chemotherapy/how-chemotherapy-drugs-work.html
- Malhotra V, Perry MC. Classical chemotherapy: mechanisms, toxicities and the therapeutic window. Cancer Biol Ther 2003;2:S2-4.
- Rang HP, Dale MM, Ritter JM, Flower RJ. Rang and dale’s Pharmacology. Philadelphia, Elsevier publisher 2007;718-38.
- World Health Organization. WHO:Introduction to drug utilization research / WHO International Working Group for Drug Statistics Methodology, WHO Collaborating Centre for Drug Statistics Methodology, WHO Collaborating Centre for Drug Utilization Research and Clinical Pharmacological Services.Geneva, WHO, 2003. Available from: http://www.whocc.no/filearchive/publications/drug_utilization_research.pdf
- Haskell CM. Principles of cancer chemotherapy. Cancer treatment 4th Edition, Philadelphia, WB Saunders 1995;31-57.
- Pirmohamed M, Park BK. Genetic susceptibility to adverse drug reactions. Trends Pharmacol Sci 2001;22:298-305.
- Khan GM, Thapa RK, Adhikari DS, Rajbhandari M, Dwa P, Shrestha S, et al. Evaluation of cancer prevalence and cytotoxic medication prescribing in central region of Nepal. Kathmandu university journal of science, engineering and technology 2013;9:189-99.
- Wooldridge JE. Corticosteroids in advancer cancer. Oncology 2001;15:225-36.
- Hesketh PJ. Defining the emetogenicity of cancer chemotherapy regimens: relevance to clinical practice. Oncologist 1999;4:191-6.
- Rodriguez M, Solanki DL, Whang R. Refractory potassium repletion due to cisplatin-induced magnesium depletion. Arch Intern Med 1989;149:2592-4.
- Lajer H, Daugaard G. Cisplatin and hypomagneÂÂsemia. Cancer Treat Rev 1999;25:47-58.
- Wolters Kluwer Health-Up-to-date. Cisplatin nephrotoxicity 2013. Available from: http://www.uptodate.com/contents/cisplatin-nephrotoxicity.
- Losonczy G, Mathe C, Muller V, Szondy K, Moldvay J. Incidence, risk factors and prevention of cisplatin induced nephrotoxicity in patients with lung cancer. Magy Onkol 2010;54:289-96.
- The Scott Hamilton CARES initiative. Heartburn and chemotherapy. Available from: http://chemocare.com/chemotherapy/side-effects/heartburn.aspx#.U0sDNPmSwd4
- Prasad A, Datta PP, Bhattacharya J, Pattanayak C, Chauhan AS, Panda P, et al. Pattern of Adverse Drug Reactions Due to Cancer Chemotherapy in a Tertiary Care Teaching Hospital in Eastern India. J Pharmacovigilance 2013;1:107-10.
- De A. Monitoring of Suspected Adverse Drug Reactions in Oncology Unit of an Urban Multispeciality Teaching Hospital. Int J Res Pharm Biom Sci 2010;1:1-32.
- Mallik S, Subish P, Ojha P, Mishra P. Pattern of adverse drug reactions due to cancer chemotherapy in a tertiary care teaching hospital in Nepal. Pak J Pharm Sci 2007;20:214-8.
- Kirthi C, Afzal A, Reddy M, Ali SA, Yerramilli A, Sharma S, et al. A study on the adverse effects of anticancer drugs in an oncology center of a tertiary care hospital.Int J Pharm Pharm Sci 2014;6:580-3.
- Poddar S, Sultana R, Sultana R, Akbor MM, Azad MAK, Hasnat A, et al. Pattern of adverse drug reactions due to cancer chemotherapy in tertiary care teaching hospital in Bangladesh. Dhaka Univ. J. Pharm. Sci 2009;8:11-16.
- Guo HJ, Ren F, Zhang D, Ji M. Monitoring report on 341 cases of adverse reactions caused by antitumor drugs. Afr J Microbiol Res 2012;30:3774-7.
- Cancer Network. Experts Debate Bolus vs Continuous Infusion 5-FU. Avilable from: http://www.cancernetwork.com
- Zaki SA. Adverse drug reaction and causality assessment scales. Lung India 2011;28:152-3.