Comorbidity-Polypharmacy Score Predicts Readmissions and in- Hospital Mortality: A Six-Hospital Health Network Experience
2 The Research Institute, The University of Toledo, College of Medicine and Life Sciences, Toledo, Ohio, USA
3 Analytics and Business Intelligence Team, The University of Toledo, College of Medicine and Life Sciences, Toledo, Ohio, USA
4 Department of Nursing, The University of Toledo, College of Medicine and Life Sciences, Toledo, Ohio, USA
5 Department of Surgery, Johns Hopkins Medicine, Baltimore, Maryland, USA
6 Department of Pharmacy, The University of Toledo, College of Medicine and Life Sciences, Toledo, Ohio, USA
7 Department of Geriatric Medicine, The University of Toledo, College of Medicine and Life Sciences, Toledo, Ohio, USA
8 Department of Surgery, The Ohio State University, College of Medicine, Columbus, Ohio, USA
9 Department of Anesthesiology, The University of Toledo, College of Medicine and Life Sciences, Toledo, Ohio, USA
10 MBA,Department od Research and Innovation, St. Luke’s University Health Network, Bethlehem, Pennsylvania, USA
Citation: Julia CT, Jill CS, Riley H, Amanda M, Dan F, Peter D, Joseph VS, Robert M, Alaa-Eldin AM, Michelle N, Susan DMB, Timothy H, Thomas JP, Stanislaw PS. Comorbidity-Polypharmacy Score Predicts Readmissions and in-Hospital Mortality: A Six-Hospital Health Network Experience. J Basic Clin Pharma. 2017;8:98-103.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@jbclinpharm.org
Abstract
Introduction: The comorbidity-polypharmacy score (CPS) is a simple sum of pre-admission medications and comorbid conditions. Previous studies show that CPS correlates with morbidity, mortality, readmissions, adverse events, and acuity level determinations in various patient populations. The aim of this study is to determine the behavior of CPS across a large sample of medicalsurgical patients, inclusive of all age ranges. We hypothesized that CPS will be associated with readmissions, mortality, and hospital length of stay. Methods: We performed an IRB-exempt study of patients admitted to a sixhospital network between Jul 2014-Dec 2014. Variables analyzed included demographics (age, gender); polypharmacy data (number of pre-admission medications); comorbid conditions (all “pre-existing” conditions on admission); hospital length of stay (HLOS); need for ICU; discharge to home versus nursing facility; and mortality. Descriptive and univariate analyses were performed using 3-point CPS increments, with mortality, readmissions, and HLOS as primary end-points. Subsequent multivariate analyses were performed for variables reaching significance level of p<0.10 in univariate analyses. SPSS 18 Statistics (IBM, Armonk, NY) was used, with statistical significance set at α < 0.01 due to multiple comparisons. Results: A total of 20,644 medical-surgical patients were studied. In univariate analyses, CPS was significantly associated with patient age, gender, length of stay, readmission, discharge destination, ICU requirement, and mortality (all, p<0.001). On multivariate analyses, factors independently associated with mortality included age (OR 1.03 per year); CPS (OR 1.05 per unit); and need for ICU (OR 21.9). Factors independently associated with readmission included age (OR 1.01 per year) and CPS (OR 1.04 per unit). ICU stay was not a predictor of readmission after correcting for index admission mortality. Conclusions: We found that CPS is independently associated with readmissions and mortality across all age groups. Further research in this area is warranted, with focus on CPS as a potential frailty indicator, as well as contributions of specific comorbidities and/or medication classes to the overall risk of mortality or readmission. CPS appears to provide a reasonable platform for patient risk stratification based on easily obtainable clinical data inputs.
Keywords
Comorbid conditions, polypharmacy, comorbiditypolypharmacy score, clinical outcomes, hospital readmissions, patient outcomes research
Introduction
The concepts of comorbidity and polypharmacy are emerging as important factors in the evaluation, risk stratification, and treatment of hospitalized patients.[1,2] With a vast increase in the proportion of elderly patients requiring care, we have seen the number of comorbid conditions and associated medications escalate significantly. Patients with high comorbidity indices tend to exhibit increased propensity toward poor outcomes, greater frequency of healthcare encounters, adverse events, and increased medical resource utilization.[3-10] However, existing clinical instruments designed to evaluate the synergistic impact of comorbidities and polypharmacy are limited by their complexity and translatability across various healthcare settings. [1,11-14] The comorbidity-polypharmacy score (CPS) is a relatively new clinical tool in the area of quantitative comorbidity assessment.[6] Easy to calculate, the CPS is derived by adding all pre-existing medical conditions and medications. By combining the number of comorbid conditions and the “intensity” or sheer number of the accompanying medical therapy, the CPS has been postulated to function as a proxy for the patient’s “physiological age”.[1] Previously published studies demonstrated a correlation between the CPS and hospital readmissions, disposition to extended care facilities, hospitalization duration metrics, adverse events/complications and mortality.[2,6-10,15]
The aim of the current study was to describe the behavior of CPS relative to a number of key clinical outcomes-including mortality, lengths of stay, readmissions, and direct costs of care-using a large, administrative dataset that incorporates all patient age groups. We hypothesized that significant correlations exist between CPS and mortality, length of stay, readmissions, and a number of secondary parameters (discharge disposition, direct admission costs) within a large administrative sample of medical-surgical patients.
Methods
This is an IRB-exempt retrospective study of medical-surgical patients admitted to a multi-hospital health network between July 1, 2014 and December 31, 2014. Study data were abstracted from an existing inpatient admission dataset generated by linking the network’s administrative and financial departments. All patients admitted to either an inpatient or observation status in any of the six hospitals within our health network during the six-month study time frame were included.
Descriptive and univariate analyses were performed using incremental 3-point CPS ranges, with 30-day mortality, 30-day readmissions, and hospital length of stay (HLOS) as primary end-points. Our secondary outcomes included hospital costs, need for ICU level of care, and postdischarge destination. Collected data included patient demographics, pre-hospital medications and comorbidities (both based on electronic medical record), in-hospital all-cause mortality, length of stay, ICU requirement, 30-day readmission, time between discharge and readmission, discharge disposition, and cost (not charge) of hospitalization.
Considering that prior studies using CPS were limited by small sample size, traditional stratification had been performed using 8-point increments.[6] With a larger sample size, the current study has allowed us to stratify CPS ranges with a much higher level of granularity (e.g., increments of 3 points). This, in turn, provided a better overall picture of CPS characteristics (e.g., the presence of any inflection points).
Characteristics of the patient sample were presented using descriptive statistics, summary tables and graphs. Normality was assessed for all continuous variables using the Kolmogorov-Smirnov test, with normally distributed data analyzed using analysis-of-variance (ANOVA) and non-normally distributed variables analyzed using the Kruskal-Wallis test. Categorical variables were analyzed using chisquare tests. Variables reaching statistical significance (p < 0.10) based on univariate analyses were subsequently included in a multivariate direct logistic regression model, with statistical significance set at α = 0.01. Finally, sensitivity analyses were performed using receiver operating curves (ROC) in order to determine which variables correlated most strongly with mortality and readmissions. All analyses were performed using SPSS version 18 (IBM Corp., Armonk, New York, USA), with key results reported as frequencies (percentages), mean ± standard deviation, median (interquartile range, IQR), and adjusted odds ratios (AOR) with 95% confidence intervals (CI). Additional data interpretation and analyses were performed at The Ohio State University, Johns Hopkins University, and The University of Toledo.
Results
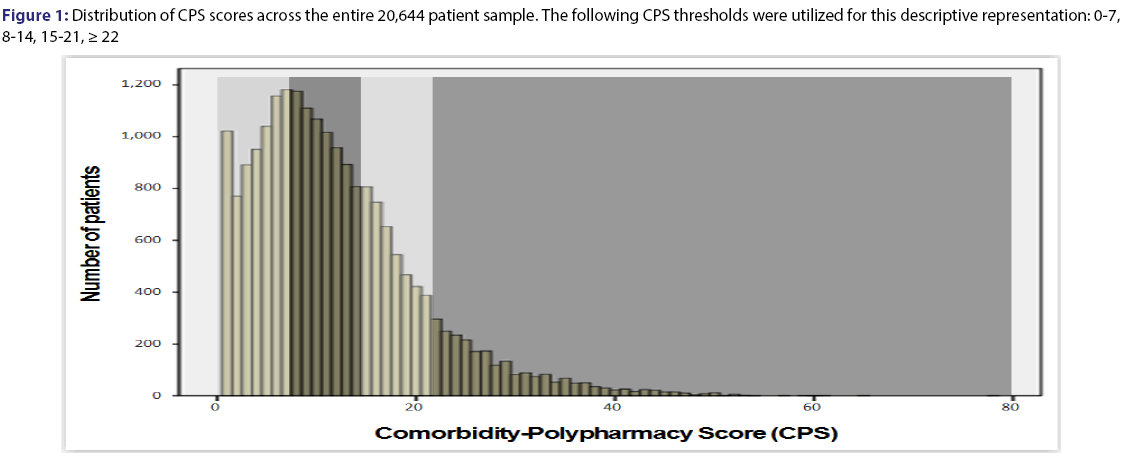
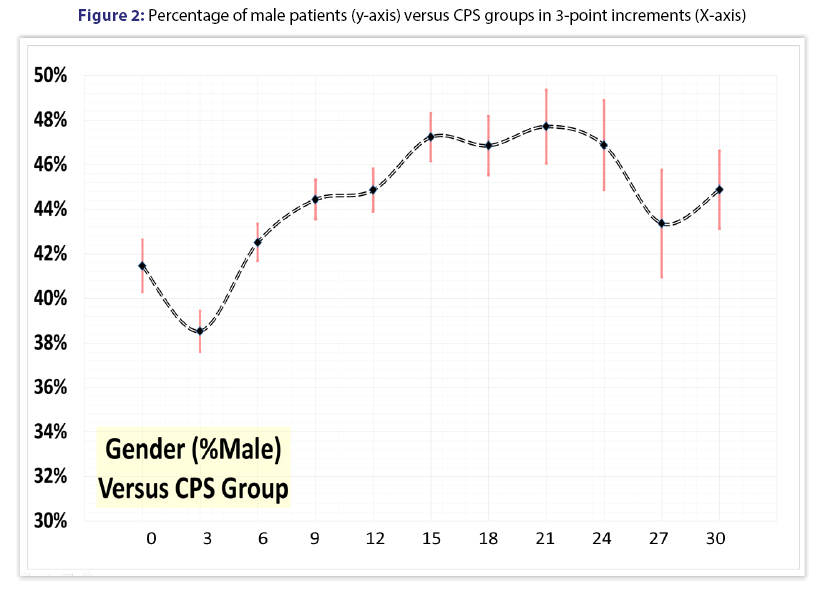
We studied 20,644 medical-surgical patients of all ages, with a mean age of 55.4 ± 26.6 years (median 60.0, IQR 36-77, range 0-115). Females constituted 56.2% (11,612/20,644) of the overall sample, with males accounting for 43.8% (9,032/20,644). Figure 1 shows the frequency distribution of CPS scores across the study sample. Figure 2 demonstrates the percentage of male patients for each 3-point CPS increment.
Mean hospitalization cost was $5,099.68 ± 7,497.65 (median $2,709.61, IQR $1,660.69-$5,705.08). Mean hospital length of stay was 4.47 ± 5.72 days (median 3, IQR 2-5, range 0-157). Reported readmission rate was 8.55% (1,765/20,644), with mean time to readmission of 12.4 ± 8.4 days (median 11.0, IQR 5-19, range 1-30). Of the patients studied, 6.14% (1,268) required ICU level of care during their hospitalization. Most patients in the current sample (76.8% or 15,846/20,644) were discharged to home, with the remainder (23.2% or 4,783/20,644) either discharged to a skilled nursing or a long term acute care facility.
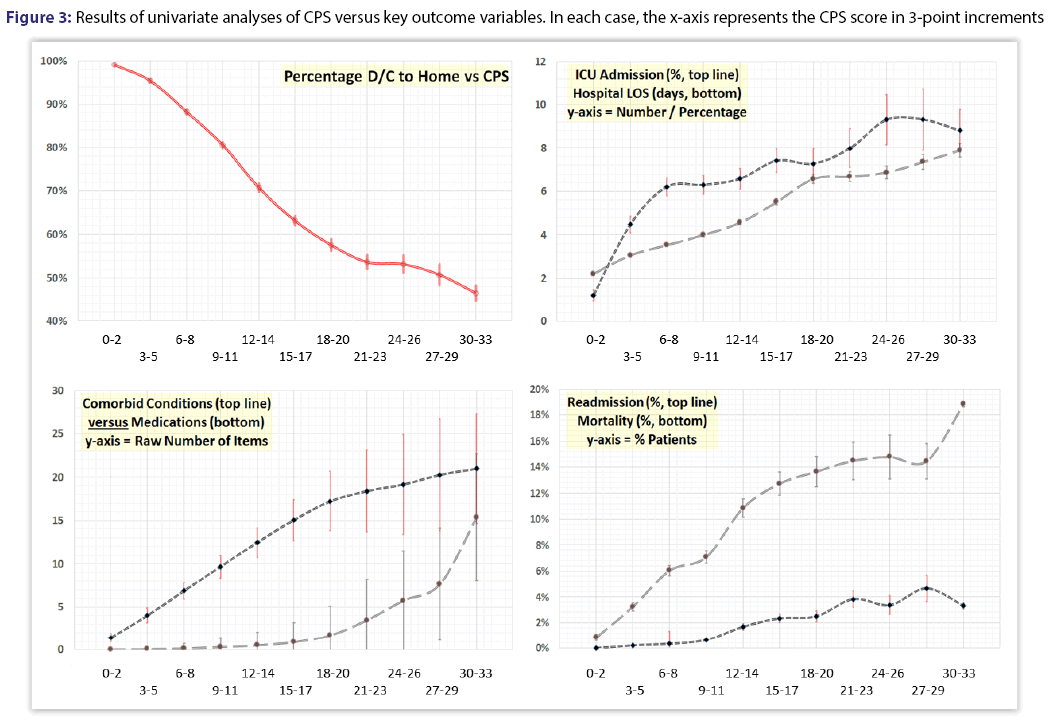
The study population had a median CPS of nearly 10. A detailed relationship between comorbid conditions and polypharmacy across the full spectrum of CPS can be seen in Figure 3 (bottom left). The initial increase in CPS is primarily attributable to comorbid conditions (top line), followed by a crescendo pattern in the number of recorded medications (bottom line).
Univariate analyses demonstrated that CPS was significantly associated with patient age and gender, as well as outcome and resource consumption measures including length of stay, readmission, discharge destination, need for ICU transfer or admission, and mortality (all, p<0.001, Figure 3). In addition, direct admission costs escalated with increasing CPS (Tables 1 and 2).
| Characteristic | Value |
|---|---|
| Age (yrs) | 55.4 ± 26.6 [median 60, IQR 36-77, range 0-115] |
| Gender (n, %) | Female 11,612 (56.2%); Male (9,032 (43.8%) |
| Hospital cost ($) | $5,099.68 ± 7,497.65 [median $2,709.61, IQR $1,660.69-$5,705.08] |
| Readmission (n, %) | 1,765/20,644 (8.55%) |
| Time to readmission (d) | 12.4±8.4 [median 11.0, IQR 5-19, range 1-30] |
| ICU required (n, %) | 1,268/20,644 (6.14%) |
| Discharge to home (n, %) | 15,846/20,644 (76.8%) |
| CPS (units) | 11.94±8.30 [median 10.0, IQR 6-16, range 1-78] |
Table 1: General characteristics of the patient sample
| CPS RCPSange | 0-2 | 3-5 | 6-8 | 9-11 | 12-14 | 15-17 | 18-20 | 21-23 | 24-26 | 27-29 | 30-33 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean cost (± Std Error) | $1,851 ± $69 |
$3,698 ± $82 |
$4,639 ± $108 |
$5,005 ± $113 |
$5,113 ± $141 |
$5,942 ± $169 |
$7,391 ± $306 |
$6,769 ±$272 |
$7,282 ± $406 |
$8.429 ± $683 |
$7,938 ±$331 |
| Median cost | $1,237 | $2,154 | $2,501 | $2,747 | $2,854 | $3,506 | $3,978 | $4,070 | $4,418 | $4,637 | $4,672 |
Table 2: Relationship between CPS range and direct admission cost (p<0.001). Both mean and median costs are shown for each CPS range
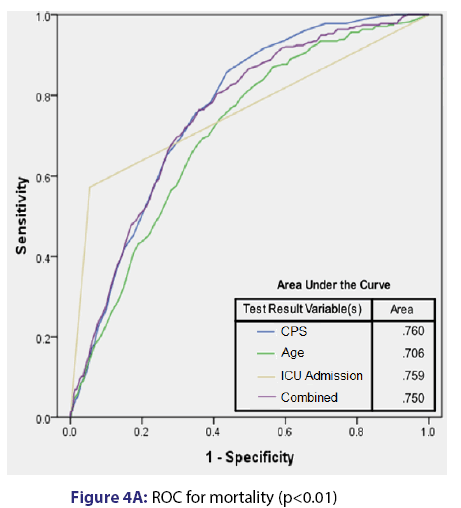
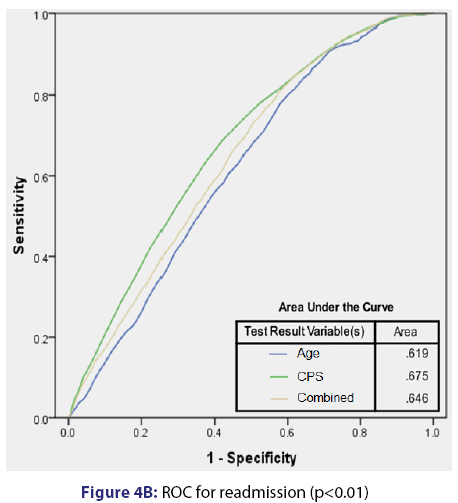
Based on multivariate analyses, CPS was independently associated with mortality (AOR 1.05 per unit) and readmission (AOR 1.04 per unit). Additionally, ICU admission (AOR 21.9) and age (AOR 1.03 per year) were independently associated with mortality. Finally, patient age (AOR 1.01) was independently associated with readmission. The summary of multivariate analysis results is provided in Table 3. Receiver operating curves (ROC) analyses [Figure 4] show that CPS provides a reasonable predictive power regarding mortality, which appeared to be more robust than age, ICU admission, and all three elements (CPS, age, and ICU admission) combined. In terms of readmission, the predictive value of CPS was not as robust, but it remained the strongest predictor. Of note, when looking at readmissions, the ROC for combined age and CPS was less predictive than CPS in isolation.
| Variable | Adjusted Odds Ratio (Mortality) | 95% Confidence Interval |
|---|---|---|
| Age | 1.028 (per year) | 1.021-1.036 |
| CPS | 1.052 (per unit) | 1.039-1.066 |
| ICU admission | 21.9 (binary) | 17.199-28.481 |
| Variable | Adjusted Odds Ratio (Readmission) | 95% Confidence Interval |
| Age | 1.012 (per year) | 1.010-1.015 |
| CPS | 1.047 (per unit) | 1.041-1.053 |
Table 3: Results of multivariate analyses. Featured are factors independently associated with mortality and hospital re-admission
To better demonstrate the utility of CPS as a predictor of mortality [Table 4] and readmission [Table 5] across all age groups (stratified by 10-year increments), we performed bivariate regression analyses for each respective end-point, using CPS and Age as comparator variables. As seen in these analyses, CPS (a surrogate for “physiological age”) is consistently a better predictor of mortality and readmission for most age-based strata.
| Age Strata | CPS [AOR, 95% C.I.] | Age [AOR, 95% C.I.] |
|---|---|---|
| 0-10 years | 1.216, 1.048-1.392* | 0.312, 0.040-2.442 |
| 11-20 years | 1.434, 0.930-2.211 | 0.793, 0.366-1.491 |
| 21-30 years | 1.150, 1.063-1.244* | 1.044, 0.733-1.488 |
| 31-40 years | 1.142, 1.061-1.230* | 0.769, 0.556-1.064 |
| 41-50 years | 1.083, 1.021-1.148* | 1.041, 0.847-1.281 |
| 51-60 years | 1.085, 1.044-1.127* | 1.049, 0.879-1.251 |
| 61-70 years | 1.064, 1.038-1.090* | 1.014, 0.920-1.116 |
| 71-80 years | 1.053,1.029-1.079* | 0.930, 0.853-1.015 |
| 81-90 years | 1.041, 1.017-1.065* | 0.977, 0.903-1.056 |
| 91+ years | 1.042, 1.003-1.083* | 1.033, 0.912-1.169 |
Table 4: Results of bivariate regression analysis for mortality, performed across 10-year strata of patient age, examining the relative contribution of CPS versus chronological age. While age was not independently associated with mortality in any of the strata, CPS was predictive in all but one stratum (11-20 years). Legend: CPS = Comorbidity-Polypharmacy Score; AOR=Adjusted Odds Ratio; 95% C.I. = 95% Confidence Interval; *=statistically significant result
| Age Strata | CPS [AOR, 95% C.I.] C.I.] |
Age [AOR, 95% |
|---|---|---|
| 0-10 years | 1.033, 0.833-1.282 | 1.286, 1.075-1.539* |
| 11-20 years | 1.163, 1.012-1.336* | 1.072, 0.788-1.459 |
| 21-30 years 31-40 years |
1.125, 1.088-1.164* 1.062, 1.029-1.095* |
1.053, 0.971-1.142 1.182, 1.084-1.289* |
| 41-50 years | 1.074, 1.053-1.097* | 1.006, 0.947-1.069 |
| 51-60 years 61-70 years |
1.052, 1.036-1.068* 1.033, 1.021-1.045* |
1.010, 0.958-1.064 1.014, 0.979-1.051 |
| 71-80 years | 1.033, 1.021-1.045* | 1.010, 0.973-1.047 |
| 81-90 years | 1.042, 1.029-1.056* | 1.061, 1.018-1.106* |
| 91+ years | 1.011, 0.983-1.039 | 1.027, 0.950-1.110 |
Table 5: Results of bivariate regression analysis for readmission, performed across 10-year strata of patient age, examining the relative contribution of CPS versus chronological age. While age was independently associated with readmission in only 3 strata (0-10, 31-40, and 81-90 years), CPS was predictive in all but two strata (0-10 and 91+ years). Legend: CPS = Comorbidity-Polypharmacy Score; AOR=Adjusted Odds Ratio; 95% C.I. = 95% Confidence Interval; *=statistically significant result
Discussion
The closely linked phenomena of polypharmacy and multi-morbidity constitute the hallmark of modern treatment of complex medical patients, regardless of chronological age or specific diagnoses.[16-18] The overarching aim of this study was to determine the behavior of CPS in a large, broadly inclusive sample of medical-surgical patients, across of all age ranges. In addition, we investigated which factors, among key outcome variables, were independently associated with 30-day patient mortality and hospital readmissions at our hospital network during the 6-month study period. Our results add significant new knowledge and understanding in all of the above domains, as outlined in the subsequent paragraphs.
In the acute setting, significant challenges emerge when managing patients with increasing physiological age and loss of the so-called “physiological reserve”, regardless of the chronological age.[1] Elevated CPS (e.g., ≥ 15) has been associated with poor hospital outcomes in older (≥ 45 years) trauma patients.[6,7,15] In addition, CPS was shown to predict readmissions in trauma patients,[2] adverse events in patients with advanced Parkinson’s disease,[10] as well as inpatient morbidity and discharge to extended care facilities in burn patients ≥ 45 years of age.[8] Central to this study’s hypothesis, we estimated that there was a significant, step-wise relationship between CPS and key clinical outcome measures across all age groups. Due to low incidence of multi-morbidity and polypharmacy among younger patients, a much larger sample was required to achieve sufficient data granularity to demonstrate the effect of increasing CPS on outcomes in patient’s ≤ 45 years of age.
Although many patients had no medications listed on initial presentation, the vast majority had at least one comorbidity present on admission Figure 1. As shown in Figure 3 (bottom left), a structured relationship can be seen between the two components within the CPS-the number of medications and the number of pre-existing comorbidities. At lower values of CPS, the relative contribution of comorbidities is greater than that of medication. This changes as the CPS mid-range is reached (approximately 15-20), at which point there is an increasingly greater relative contribution of medications to the overall score. This trend strongly suggests that that the number of pharmaceuticals being co-administered at any given level of comorbidity corresponds to the overall “intensity” of the medical management required to control the corresponding comorbid conditions.[1] Given the close inter-relationship between the above concepts, the combined comorbidity-polypharmacy assessment could provide an indirect measure of the physiological age, and possibly even frailty.[19] However, further validation is required to prove these as yet hypothetical relationships.
The relationship between CPS and hospital readmissions is important in the era of healthcare cost containment and pay-for-performance measures.[20,21] Our analyses show that CPS is independently associated with 30-day readmissions. More specifically, we noted a 9-fold increase in readmissions between the lowest and the highest CPS ranges (Figure 3, bottom right). This, in turn, supports the use of CPS as a stratification tool for determining readmission risk and corroborates previously published data while extending the applicability of CPS to medical-surgical patients of all age groups.[2]
We found that CPS significantly correlated with both financial and non-financial measures of healthcare resource consumption. This is supported by trends evident in four key parameters-hospital length of stay, hospitalization costs, proportion of patients admitted to ICU, and the proportion of patients discharged to skilled nursing facility (SNF). Correlations between increasing CPS, hospital lengths of stay, proportion of patients admitted to ICU, and the greater need for discharge to SNF have been previously reported for older patient populations.[1,6-8,15] The unique character of the current study is that it extends these findings to all age groups and enables generalization of the results to all medical-surgical patients. This is also the first study demonstrating that CPS increases correlate with escalating admissionrelated costs (as opposed to charges, Table 2).
This study has several potential limitations, beginning with the retrospective nature of the data and the reduced number of parameters available for analysis. As with any retrospective analysis, only phenomenological observations can be made, rather than determinations of causation. Our ability to extract clinical information from various administrative and financial data sets was also restricted. For example, lack of physiologic acuity information precludes patient stratification when performing mortality analyses based on ICU admission. Our dataset did not include factors such as race, ethnicity, and/or socio-economic status. Both medical records and claims data may be incomplete and are prone to error, creating a potential source bias. Despite that, our results are generally consistent with our previous research in this area, majority of which was performed at other locations and using different data collection paradigms. It is also important to note that this study might not be broadly generalizable as it was done at a single health network. Although our network encompasses six hospitals, a high degree of standardization between individual locations makes the translatability of our data difficult outside of networks and/ or hospitals of similar general characteristics and size. A future multicenter study expanding beyond our network may help alleviate some of these concerns. Advantages of the current study include a large sample size as well as high quality of source information included within the limited dataset.
Previous investigations have proposed using CPS to assist in the triage of trauma patients to either medical-surgical or ICU level of care.[9] We propose further studies in which CPS, along with severity of illness/ injury, is evaluated as a tool for triaging patients to an appropriate level of inpatient care. Although yet to be demonstrated empirically, we believe that CPS may be a surrogate measure of frailty, and its correlation with both mortality and readmission can help guide risk stratification and resource allocation considerations. Future studies correlating CPS with frailty should incorporate the concept of the “frailty phenotype”[22] and the frailty index.[23] An optimal frailty assessment should be able to measure the patient’s capacity to “absorb and withstand” physiologic stress.[24] Results of the current study, combined with previous CPS research,[1,2,6,8,9] suggest that the ability of the CPS to better quantify the “intensity” of medical therapy may represent an important link between the concepts of comorbidity and frailty.[1,6,25]
Conclusion
This study demonstrates that CPS is independently associated with risk of readmission and mortality across all age groups. Our results also show that CPS may be considered a predictor of healthcare resource consumption, given its significant correlations with hospital lengths of stay and hospitalization costs. Further research in this area is warranted, focusing on the evaluation of CPS as a potential frailty indicator, the determination of relative contributions of specific comorbidities and/ or medication classes to the overall risk of mortality or readmission, and dedicated analyses examining the role of CPS as a predictor of healthcare resource consumption. CPS appears to provide a reasonable platform for patient risk stratification based on readily obtainable clinical and administrative data inputs.
Conflict of Interest Statement
The authors declare no conflict(s) of interest related to this scholarly work.
Source of funding statement
This project was not externally funded and was completed independently by the authors.
REFERENCES
- Stawicki SP. Comorbidity polypharmacy score and its clinical utility: A pragmatic practitioner's perspective. J Emerg Trauma Shock 2015;8:224-31.
- Housley BC. Comorbidity-polypharmacy score predicts readmission in older trauma patients. J Surg Res 2015;199:237-43.
- Karunananthan S. A multidisciplinary systematic literature review on frailty: overview of the methodology used by the Canadian Initiative on Frailty and Aging. BMC Med Res Methodol 2009;9:68.
- Walston J. Research agenda for frailty in older adults: toward a better understanding of physiology and etiology: summary from the American Geriatrics Society/National Institute on Aging Research Conference on Frailty in Older Adults. J Am Geriatr Soc 2006;54:991-1001.
- Clegg A. Frailty in elderly people. Lancet 2013;381:752-62.
- Evans DC. Comorbidity-polypharmacy scoring facilitates outcome prediction in older trauma patients. J Am Geriatr Soc 2012;60:1465-70.
- Evans DC. Pre-injury polypharmacy as a predictor of outcomes in trauma patients. Int J Crit Illn Inj Sci 2011;1:104-9.
- Justiniano CF. Comorbidity-polypharmacy score predicts in-hospital complications and the need for discharge to extended care facility in older burn patients. J Burn Care Res 2015; 36:193-6.
- JustinianoCF. Comorbidity-polypharmacy score: a novel adjunct in post-emergency department trauma triage. J Surg Res 2013;181:16-9.
- Cheron J. The use of enteral access for continuous delivery of levodopa-carbidopa in patients with advanced Parkinson’s disease. United European Gastroenterology Journal 2017;5:60-68.
- Bouillon K. Measures of frailty in population-based studies: an overview. BMC Geriatr 2013;13:64.
- Ladha KS. The Deyo-Charlson and Elixhauser-van Walraven Comorbidity Indices as predictors of mortality in critically ill patients. BMJ Open 2015;5:p. e008990.
- Menendez ME. Predicting In-Hospital Mortality in Elderly Patients With Cervical Spine Fractures: A Comparison of the Charlson and Elixhauser Comorbidity Measures. Spine (Phila Pa 1976) 2015;40:809-15.
- Menendez ME. The Elixhauser comorbidity method outperforms the Charlson index in predicting inpatient death after orthopaedic surgery. Clin Orthop Relat Res 2014;472:2878-86.
- Mubang RN. Comorbidity-Polypharmacy Score as Predictor of Outcomes in Older Trauma Patients: A Retrospective Validation Study. World J Surg 2015;39:2068-75.
- Birriel TJ. Adverse drug reactions in the era of multi-morbidity and polypharmacy. J Basic Clin Pharm 2015;6:122-3.
- Fabbri E. Aging and multimorbidity: new tasks, priorities, and frontiers for integrated gerontological and clinical research. Journal of the American Medical Directors Association 2015;16:640-7.
- Jacobs J, Fisher P. Polypharmacy, multimorbidity and the value of integrative medicine in public health. European Journal of Integrative Medicine 2013;5:4-7.
- Cohen MS. Patient frailty: Key considerations, definitions, and practical implications. 2016.
- Kahn CN. Assessing Medicare’s hospital pay-for-performance programs and whether they are achieving their goals. Health Affairs 2015;34:1281-8.
- Figueroa JF, Wang DE, Jha AK. Characteristics of hospitals receiving the largest penalties by US pay-for-performance programmes. BMJ quality & safety 2016:bmjqs-2015-005040.
- Fried LP. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001;56:M146-56.
- Rockwood K. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005;173:489-95.
- Cesari M. The frailty phenotype and the frailty index: different instruments for different purposes. Age Ageing 2014;43:10-2.
- Searle SD. A standard procedure for creating a frailty index. BMC Geriatr 2008;8:24.