Assessment of Knowledge, Attitude and Practice of Pharmacy Professionals Toward Generic Medicines, Northern Ethiopia, Mekelle: A Cross Sectional Study
2 Lecturer at Addis Ababa University School of Pharmacy, Addis Ababa, Ethiopia
3 Forecasting and capacity building officer at the federal democratic republic of Ethiopia Pharmaceutical fund and supply agency, Addis Ababa, Ethiopia
Citation: Yard B. Assessment of Knowledge, Attitude and Practice of Pharmacy Professionals Toward Generic Medicines, Northern Ethiopia, Mekelle: A Cross Sectional Study. J Basic Clin Pharma 2017;8:193-199.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@jbclinpharm.org
Abstract
Introduction: Generic drugs have the same chemical formula as brand product and they are bioequivalent to the originator product. Insufficient knowledge of healthcare professionals about generics causes’ reduced utilization of these drugs. Therefore, this study narrows the knowledge gap and the findings assist policy makers regarding generic prescribing in the future. Aim: To assess knowledge, Attitude and practiceof pharmacy professionals toward generic products. Method: The study was conducted in all medicine retail outlets in Mekelle city, from April 10, 2016-May 12, 2016. Facility based cross -sectional study was used and the data collection tool were self-administered questionnaire. Statistical test binary logistic regression has been done to determine predictors of knowledge and attitude toward generic medicines. Results: From the current study more than half of the respondents 52.9% (agree=32.2%, 20.7% strongly agree) claimed that they have had knowledge on the concept of generic medicine. Pharmacy professionals with experience of from 2 to 5 years (AOR=25.620 [1.954-335.896) and those with more than 5 years (AOR=106.543 [2.375-4779.542) were more likely to have positive attitude toward generic medicines compared to those with work experience of up to 2 years. Conclusion: More than half of the study participants have had knowledge on the concept of generic medicine and their right to perform generic substitution. The professionals with more experience in the sector could have better view on generic product.
Keywords
Brand, generic; mekelle, pharmacy professionals
Introduction
Generic drugs have the same chemical formula as brand product,[1] contains the same active substance as the reference medicine, and it is used at the same dose(s) to treat the same disease as the reference medicine.[2] A generic medicine is a multisource pharmaceutical product which is meant to be interchangeable [3,4] with the comparator product also called innovator, proprietary or brand product. Interchangeable pharmaceutical products are considered as therapeutically equivalent of an innovator product [5]. In order to be approved, a generic medicine should be bioequivalent to the originator product, and must be the same in terms of strength, safety and quality;[3,4,6] they must be able to demonstrate bioequivalence to the originator medicine in terms of bioavailability and the rate of absorption.[7] However, the name of the medicine, its appearance (such as color or shape), its packaging,[2,7,8] size and excipient ingredients [7] can be different from those of the reference medicine. Pre-clinical and clinical testing did not have to be repeated again for generics. The deliberate result of this legislation was to guarantee that generic medicines would be cheaper than the equivalent originator medicine because it was not expected for generic medicine manufacturers to repeat again innovation, pre-clinical and clinical studies.[2]
Globally, the use of generic drugs has increased gradually as a result of financial pressure on drug budgets. In several countries throughout the globe, the practice of generic substitution (GS) is strongly supported by health authorities.[9-14] Public and private third-party payers and health care regulatory authorities increasingly encourage or obligate the use of generics through measures such as generic prescribing and generic substitution.[15-19] In Ethiopia, clinical practice guidelines enforce a prescription to be using generic names of the medicine(s).[20] In addition, the national drug policy offers a pharmacist the right to dispense generic drugs as substitutes for prescribed brand names.[21]
The concepts of generic prescribing and generic replacement have been controversial, and questions continue concerning the acceptance and promotion of generic medicines among healthcare professionals.[22-25] In the UK, for example, there was strong opposition when plans were anticipated to introduce generic replacement into UK primary care.[26] This controversy surrounds issues of quality, safety and efficacy of the generic medicines.[23,24,27,28] There is an apparent lack of knowledge pertaining to the efficacy, safety and quality of generic drugs among healthcare professionals.[29] It is imperative analyzing of the perception and attitude of participating stakeholders regarding generic medicines is a pre requisite to promote the utilization of generics.[5,28]
Inaccurate or insufficient knowledge of healthcare professionals about generics causes’ hesitation on the utilization of these drugs and chiefly about their efficacies and this is becoming a major obstacle to a wider use of these products.[4,30] Studies on consumers’ perceptions suggest that pharmacists are main determinants in consumers’ choice to rational use of generics.[23,30] A study in the area employed pharmacist and physician depicted 51.1% of the respondents believed as generic and brand drugs are totally similar. In the study 59.2% of them disagreed with the concept generic drugs have more side effects than brand drugs.[30] This study narrows the knowledge gap and the findings assist policy makers regarding generic prescribing in the future. The main
Methods
Study area and period
The study was conducted in all medicine retail outlets in Mekelle city. Mekelle is the capital city of Tigray regional state, around 783 km away from Addis Ababa the capital city of Ethiopia. In Mekelle there are 2 governmental general hospitals, 1 comprehensive specialized hospital, 32 pharmacies, 47 drug stores and 3 drug venders. This study was conducted from April 10, 2016-May 12, 2016.
Study design
Facility based cross-sectional survey was used to assess Knowledge, Attitude and Practice of Pharmacy Professionals towards generic medicines at Mekelle city, Northern Ethiopia.
Population
Source population
The source population was all pharmacy professional work in private retail outlets.
Study population
Those pharmacy professionals working in private retail outlets and available during data collection period.
Inclusion criteria
Working in the MROs.Study variables
Study variables
Independent variables
Socio-demographic characteristics of professionals (Gender, Age, Qualification, Employment position, Education institution Membership to Ethiopian Pharmaceutical Association (EPA)Work experience (years)Monthly salary Number of prescriptions filled per day).
Dependent variables
Knowledge of community pharmacist’s on generic medicine.Attitude toward generic medicine and practice regarding generic product.
Data collection and management
A self-administered questionnaire has been developed through an extensive review of available literature on knowledge, attitudes and practices of pharmacists regarding generic medicines. The questionnaire was developed in English and contained parts on socio-demographic profiles of the participants which followed by questions that assess their knowledge, attitude and practice toward generic medicines. Pretest of the questionnaires were done on private community pharmacies at Quiha town which is in nearest distance to Mekelle and some of the questions were modified accordingly and the revised version of the questionnnaeire were used as a final data collection tool. Data collection was conducted from April 10, 2016 May 12, 2016 by five data collectors. Training focused on data abstraction was provided for the data collectors by principal investigator. Questionnaires were distributed and collected after five days.
Data entry, analysis and interpretation
The data collected were entered into SPSS Version 23, cleaned and analyzed. In the analysis two knowledge categories were created using the total score for knowledge questions. This ranged from 8 to 40 and a total score of more than 24 (average score) was considered to have ‘adequate knowledge’ while score less than that was considered ‘inadequate’. As to attitude, a score range of 9 to 45 was categorized using 27 as the cutoff point to delineate between positive attitude and negative attitude towards generic medicines. In the scoring, negatively worded questions were reversed. Descriptive aspects of analyses were done through calculating frequency, mean and standard deviations. Statistical test binary logistic regression has been done to determine predictors of knowledge and attitude toward generic medicines. In interpreting these tests p-value<0.05 at 95% CI was used as a cutoff point to determine statistical significance of associations.
Results
Socio-demographic and work profile
Of 100 questionnaires distributed, 89 of the participants returned the surveys, which make a response rate of 89%. From the participants in this study above a half was male and 58.2% were in the age range of 20-29. Majority of the participants (75.6%) were not the member of EPA and 63.6% of the participants were educated in private colleges [Table 1].
| Variable | N | Category of hospital Intervention (%) |
|
|---|---|---|---|
| Gender | Male | 89 | 47 (52.8) |
| Female | 42 (47.2) | ||
| Age group (years) | 20-29 | 79 | 46 (58.2) |
| 30-39 | 18 (22.8) | ||
| 40+ | 15 (19.0) | ||
| Qualification | Diploma | 89 | 34 (38.2) |
| Degree | 19 (21.3) | ||
| MSc | 36 (40.4) | ||
| Employment position | Full/part owner | 86 | 39 (45.3) |
| Employee | 46 (53.5) | ||
| Others* | 1 (1.2) | ||
| Education institution | Private institution | 88 | 56 (63.6) |
| Government/public institution | 32 (36.4) | ||
| Membership to Ethiopian Pharmaceutical Association (EPA) | Yes | 86 | 21 (24.4) |
| No | 65 (75.6) | ||
| Work experience (years) | Up to 2 | 87 | 30 (34.5) |
| From 2.1 to 5 | 37 (42.5) | ||
| More than 5 | 20 (23.0) | ||
| Monthly salary | Up to 2500 | 60 | 35 (58.3) |
| 2501 to 5000 | 20 (38.3) | ||
| More than 5000 | 5 (8.3) | ||
| Number of prescriptions filled per day | Up to 10 | 81 | 44 (54.3) |
| 11-20 | 17 (21.0) | ||
| 21-30 | 8 (9.9) | ||
| >30 | 12 (14.8) | ||
Table 1: Socio-demographic profile of community pharmacists in Mekelle city,August 2016
Knowledge of pharmacy professionals on generic medicines
From the study participants 52.9% (agree=32.2%, 20.7% strongly agree) have had knowledge on the concept of generic medicine is bioequivalent to brand medicine and 38.6% of the participants strongly agree that generic medicine must contain the same amount of active ingredients as the brand medicine. From the participants 44.2% did not support the idea that substitution of medicines with narrow therapeutic index is inappropriate [Table 2].
| Variable | N | Strongly disagree (%) | Disagree (%) | Neutral (%) | Agree (%) | Strongly agree (%) |
|---|---|---|---|---|---|---|
| A generic medicine is bioequivalent to brand medicine | 87 | 9 (10.4) | 17 (19.5) | 15 (17.2) | 28 (32.2) | 18 (20.7) |
| A generic medicine must contain the same amount of active ingredients as the brand medicine | 88 | 5 (5.7) | 10 (11.4) | 6 (6.8) | 33 (37.5) | 34 (38.6) |
| A generic medicine must be in the same dosage form as the brand medicine | 87 | 9 (10.4) | 15 (17.2) | 7 (8.1) | 35 (40.2) | 21 (24.1) |
| Generic medicines are cheaper than brand name medicines | 88 | 9 (10.2) | 9 (10.2) | 5 (5.7) | 32 (36.4) | 33 (37.5) |
| Wider use of generic medicines in Ethiopia helps in decreasing the health care expenditure of government | 87 | 10 (11.5) | 24 (27.6) | 8 (9.2) | 26 (29.9) | 19 (21.8) |
| Community pharmacists in Ethiopia have the right to perform generic substitution | 88 | 7 (8.0) | 12 (13.6) | 17 (19.3) | 32 (36.8) | 20 (23.3) |
| Substitution of medicines with narrow therapeutic index is inappropriate | 86 | 20 (23.3) | 18 (20.9) | 13 (15.1) | 25 (29.1) | 10 (11.6) |
| In Ethiopia generic medicines are approved by FMHACA just like brand medicines | 86 | 9 (10.5) | 8 (9.3) | 11 (12.8) | 40 (46.5) | 18 (20.9) |
Table 2: Knowledge of community pharmacist’s on generic medicine in Mekelle city, August 2016
From this study, Only position (owner vs employee) had statistically significant association in bivariate analysis which was lost in adjusted ORand it showed employee has less knowledge than Full/partial ownership (COR0.736[95% CI,0.150-0.946]). No significant differences were observed among other socio-demographic and work profiles on knowledge of generic medicine [Table 3].
| Variable | Knowledge on generics | Crude OR [95% CI] | Adjusted OR [95% CI] | |
|---|---|---|---|---|
| Yes (%) | No (%) | |||
| Sex | ||||
| Male | 33 (70.2) | 14 (29.8) | 1 | 1 |
| Female | 23 (54.8) | 19 (45.2) | 0.514 [0.215-1.228] | 0.761 [0.144-4.029] |
| Age group (yrs) | ||||
| 20-29 | 29 (63.0) | 17 (37.0) | 1 | 1 |
| 30-39 | 12 (66.7) | 6 (33.3) | 1.172 [0.372-3.697] | 1.231 [0.093-16.325] |
| 40+ | 8 (53.3) | 7 (46.7) | 0.670 [0.206-2.176] | 0.356 [0.017-7.543] |
| Qualification | ||||
| Diploma | 29 (56.9) | 22 (43.1) | 1 | 1 |
| Degree and above | 28 (71.8) | 11 (28.2) | 1.931 [0.792-4.706] | 1.264 [0.119-13.386] |
| Position | ||||
| Full/partial ownership | 29 (74.4) | 10 (25.6) | 1 | 1 |
| Employee | 24 (52.2) | 22 (47.8) | 0.736 [0.150-0.946]* | 0.293 [0.051-1.704] |
| Work experience | ||||
| Up to 2 | 19 (63.3) | 11 (36.7) | 1 | 1 |
| From 2.1 to 5 | 24 (64.9) | 13 (35.1) | 1.069 [0.392-2.915] | 2.257 [0.363-14.034] |
| More than 5 | 11 (55.0) | 9 (45.0) | 0.708 [0.224-2.240] | 0.908 [0.053-15.652] |
| Salary (ETB) | ||||
| Up to 2500 | 19 (54.3) | 16 (45.7) | 1 | 1 |
| 2501 to 5000 | 14 (70.0) | 6 (30.0) | 1.965 [0.613-6.298] | 4.411 [0.577-33.712] |
| More than 5000 | 3 (60.0) | 2 (40.0) | 1.263 [0.187-8.520] | 2.738 [0.099-75.797] |
| Education institution | ||||
| Private | 36 (64.3) | 20 (35.7) | 1 | 1 |
| Government | 19 (59.4) | 13 (40.6) | 0.812 [0.333-1.982] | 0.215 [0.026-1.766] |
| Membership to EPA | ||||
| Yes | 10 (47.6) | 11 (52.4) | 1 | 1 |
| No | 44 (67.7) | 21 (32.3) | 2.305 [0.846-6.276] | 6.601 [0.957-45.559] |
| Mean number of prescriptions filled per day (SD) | 1.94 (1.15) | 1.71 (1.04) | 1.217 [0.797-1.858] | 1.179 [0.458-3.036] |
Table 3: Predictors of knowledge of community pharmacist in Mekelle city, August 2016
Attitude and practice of pharmacy professionals toward generic medicines
From the respondents 34.4% has believed medicines are less effective compared to brand name medicines. However, 40.2% couldn’t believe in the idea of brand name medicines are of higher quality compared to generic drugs. From the participants 68.8% of them believed that patients should be given enough explanations about the reasons for choosing generic medicines [Table 4].
| Variable | N | Strongly disagree (%) | Disagree (%) | Neutral (%) | Agree (%) | Strongly agree (%) |
|---|---|---|---|---|---|---|
| Generic medicines are less effective compared to brand name medicines | 90 | 24 (26.7) | 21 (23.3) | 14 (15.6) | 27 (30.0) | 4 (4.4) |
| Brand name medicines are of higher quality compared to generic drugs | 89 | 18 (20.2) | 18 (20.2) | 7 (7.9) | 33 (37.1) | 13 (14.6) |
| Generic drugs produce more side effects than brand name medicines | 89 | 27 (30.3) | 21 (23.6) | 9 (10.1) | 26 (29.2) | 6 (6.7) |
| Generic medicines take longer time to give response | 88 | 16 (18.2) | 25 (28.4) | 13 (14.8) | 25 (28.4) | 9 (10.2) |
| I support generic substitution for brand name medicines in all cases where s generic is available | 87 | 14 (16.1) | 26 (29.9) | 13 (14.9) | 22 (25.3) | 12 (13.8) |
| Price difference between generic and brand medicine is often so great that I feel I must dispense prescriptions with generic substitution especially for people who do not afford. | 89 | 6 (6.7) | 10 (11.2) | 20 (22.5) | 30 (33.7) | 23 (7.7) |
| Patients should be given enough explanations about the reasons for choosing generic medicines | 90 | 4 (4.4) | 15 (16.7) | 9 (10.0) | 34 (37.7) | 28 (31.1) |
| The intensity of promotional activities by promoters plays an important role in dispensing brand medicines | 88 | 5 (5.7) | 12 (13.6) | 7 (8.0) | 34 (38.6) | 30 (34.1) |
| Community pharmacists should be allowed to perform generic substitutions without consulting prescribing physicians | 87 | 11 (12.6) | 19 (21.8) | 13 (14.9) | 25 (28.7) | 19 (21.8) |
Table 4: Attitude of community pharmacist toward generic medicine in Mekelle city, August 2016
Pharmacy professionals with experience of from 2 to 5 years (AOR=25.620 [1.954-335.896]) and those with more than 5 years (AOR=106.543 [2.375-4779.542) were more likely to have positive attitude toward generic medicines compared to those with work experience of up to 2 years [Table 5].
| Variable | Attitude toward generic | Crude OR [95% CI] | Adjusted OR [95% CI] | |
|---|---|---|---|---|
| Yes (%) | No (%) | |||
| Sex | ||||
| Male | 34 (72.3) | 13 (27.7) | 1 | 1 |
| Female | 25 (59.5) | 17 (40.5) | 0.562 [0.231-1.366] | 0.337 [0.055-2.054] |
| Age group (yrs) | ||||
| 20-29 | 27 (58.7) | 19 (41.3) | 1 | 1 |
| 30-39 | 13 (72.2) | 5 (27.8) | 1.830 [0.558-5.994] | 0.642 [0.039-10.634] |
| 40+ | 11 (73.3) | 4 (26.7) | 1.935 [0.535-7.003] | 3.337 [0.077-144.185] |
| Qualification | ||||
| Diploma | 29 (56.9) | 22 (43.1) | 1 | 1 |
| Degree &above | 30 (76.9) | 9 (23.1) | 2.529 [0.999-6.399] | 6.762 [0.347-131.611] |
| Position | ||||
| Full/partial ownership | 30 (76.9) | 9 (23.1) | 1 | 1 |
| Employee | 27 (58.7) | 19 (41.3) | 0.426 [0.165-1.100] | 1.469 [0.174-12.376] |
| Work experience (yrs) | ||||
| Up to 2 | 15 (50.0) | 15 (50.0) | 1 | 1 |
| From 2.1 to 5 | 27 (73.0) | 10 (27.0) | 2.700 [0.974-7.481] | 25.620 [1.954-335.869]* |
| More than 5 | 14 (70.0) | 6 (30.0) | 2.333 [0.707-7.704] | 106.543 [2.375-4779.542]* |
| Salary (ETB) | ||||
| Up to 2500 | 21 (60.0) | 14 (40.0) | 1 | 1 |
| 2501 to 5000 | 13 (65.0) | 7 (35.0) | 1.238 [0.396-3.875] | 0.843 [0.053-13.407] |
| More than 5000 | 2 (40.0) | 3 (60.0) | 0.444 [0.066-3.010] | 0.014 [0.000-1.351] |
| Education institution | ||||
| Private | 34 (60.7) | 22 (39.3) | 1 | 1 |
| Government | 23 (71.9) | 9 (28.1) | 1.654 [0.647-4.228] | 1.914 [0.158-23.224] |
| EPA Membership | ||||
| Yes | 10 (47.6) | 11 (52.4) | 1 | 1 |
| No | 46 (70.8) | 19 (29.2) | 2.663 [0.970-7.308] | 13.744 [0.857-220.381] |
| Mean number of prescriptions filled per day (SD) | 1.89 (1.16) | 1.79 (1.03) | 1.088 [0.713-1.661] | 0.748 [0.248-2.255] |
| Knowledge on generics | ||||
| Yes | 39 (68.4) | 18 (31.6) | 1 | 1 |
| No | 20 (60.6) | 13 (39.4) | 1.408 [0.576-3.444] | 5.066 [0.547-46.886] |
Table 5: Predictors of attitude of community pharmacist in Mekelle city, August 2016
Nearly half of the participants (48.3%) declared that lack of belief in generic medicines as important factor that hinder dispensing of generic medicine. From the participants 73.6% and 70.9% of them revealed that affordability to the customer and consumer preference as factor to reduce selection and dispensing of generic medicine [Table 6].
| Variable | N | Not important (%) | Neutral (%) | Important (%) |
|---|---|---|---|---|
| Lack of belief in generic medicines | 87 | 27 (31.0) | 18 (20.7) | 42 (48.3) |
| Availability of policies, laws ®ulations | 87 | 22 (25.3) | 29 (33.3) | 36 (41.4) |
| Affordability to the customer | 87 | 11 (12.6) | 12 (13.8) | 64 (73.6) |
| Lacking options | 85 | 30 (35.3) | 12 (14.1) | 43 (50.6) |
| Consumer preference/ demand | 86 | 13 (15.1) | 12 (14.0) | 61 (70.9) |
| Cost effectiveness of generic medicines | 87 | 19 (21.8) | 11 (12.6) | 57 (65.5) |
| Substitution agreement with prescriber | 87 | 25 (28.7) | 16 (18.4) | 46 (52.9) |
Table 6: Possible influencing factors related to selection and dispensing of generic medicines among the community pharmacists.
From this study 55.9% (agree=17%, strongly agree 38.9%) of the participant claimed locally manufactured generics are equal in their quality compared to the imported generics and 60.2% believed they are equal in their safety and efficacy. From respondents 62.4%claimed locally manufactured generics are cheaper compared to imported generics [Table 7].
| Variable | N | Strongly disagree (%) | Disagree (%) | Neutral (%) | Agree (%) | Strongly agree (%) |
|---|---|---|---|---|---|---|
| Locally manufactured generics are equal in their quality compared to the imported generics | 88 | 8 (9.1) | 17 (19.3) | 13 (14.8) | 15 (17.0) | 35 (38.9) |
| Locally manufactured generics are equal in their safety and efficacy compared to the imported generic | 88 | 7 (8.0) | 13 (14.8) | 15 (17.0) | 12 (13.6) | 41 (46.6) |
| Manufacturers of local generic products have a reliable logistic and supply system | 87 | 8 (9.2) | 12 (13.8) | 26 (29.9) | 16 (18.4) | 25 (28.7) |
| I prefer to stock and dispense locally manufactured generics because the companies provide good bonus scheme compared to suppliers importing them | 85 | 20 (23.5) | 29 (34.1) | 17 (20.0) | 9 (10.6) | 10 (11.8) |
| Credibility of the generic manufactures/suppliers is my concern when stocking medicines in my pharmacy. | 83 | 9 (10.8) | 25 (30.1) | 21 (25.3) | 11 (13.3) | 17 (20.5) |
| I will only stock locally manufactured product which is well advertised through medical representatives and medicine related references. | 86 | 13 (15.1) | 34 (39.5) | 11 (12.8) | 17 (19.8) | 11 (12.8) |
| Imported generics need to pass more stringent approval process compared with locally manufactured ones. | 86 | 7 (8.1) | 17 (19.8) | 21 (24.4) | 18 (20.9) | 23 (26.7) |
| Locally manufactured generics are cheaper compared to imported generics. | 85 | 3 (3.5) | 19 (22.4) | 10 (11.8) | 23 (27.1) | 30 (35.3) |
| Drug Regulatory Authorities need to convince pharmacists that registered locally manufactured generics are of high quality and standards. | 83 | 13 (15.7) | 19 (22.9) | 12 (14.5) | 19 (22.9) | 20 (24.1) |
Table 7: Views toward locally produced generic medicines among community pharmacists in Mekelle city, August 2016
Discussion
This study attempted to assess knowledge, attitude and practice of community pharmacist toward generic medicines and it tried to assess their views toward locally produced generic medicine in Mekelle city. From the current study more than half of the respondents 52.9% (agree=32.2%, 20.7% strongly agree) claimed that they have had knowledge on the concept of generic medicine is bioequivalent to brand medicine which is much lower than the study reported from new Zeeland which depicted 70 percent of the participants had knowledge on bioequivalence of generic medicine to the brand product; this difference might be the result of difference in educational status of the participants.[31] On the other hand the finding of this study is higher than the report of Malaysia that revealed 50.2% of the pharmacists claimed that generic medicines are therapeutically equivalent with that of innovator product.[32] From the participants 64.3% of them claimed that generic medicine must be in the same dosage form as the brand medicine which is lower finding when compared with the report of Australia that revealed 84.1% of the respondents claimed this truth. From the current study 73.9% of them claimed that generic medicines are cheaper than brand name medicines which are still lower than the report of Australia that announced 91.3% of the participants claimed generic as cheaper product than brand.[33] From the participants 59.8% of the pharmacists have had knowledge on the right to perform generic substitution which is much lower than the report from survey of 1,000 French community pharmacists that reported 90% of the pharmacists were comfortable to utilize their substitution right.[34]
From this study, Only position (owner versus employee) had statistically significant association in bivariate analysis which was lost in adjusted OR and it showed employee has less knowledge than Full/ partial ownership (COR 0.736 [95% CI, 0.150-0.946]) this difference may results from the difference in interest. Those business holders might have great interest toward their professional practice due to their From the study participant’s 34.4% have an attitude of generic medicines are less effective compared to brand name medicines and 50% of respondents disagree with the concept of generic is less effective in comparison to brand products. Study from Malaysia reported 58.4% of the pharmacist’s perceived local generics as equal in safety and efficacy as the imported products,[35] which shows better perception in generic medicine acceptance in Malaysia than Mekelle so, promotion of generic medicine might be critical work in Mekelle. More than half (51.7%) of the participants support the idea of brand name medicines are of higher quality compared to generic drugs this finding is better when we compare to the result reported from Australia that depicted 89.6% of participants claimed generic medicines are of inferior quality to brand-name drugs.[33] The difference of those two findings might be the result of the difference in the nature of the participant’s in case of Australian study, the participants were pre-registrant pharmacist and in case of our study, the practitioner from junior to senior were involved. From the current study 39.1% of the respondent’s claimed that they support generic substitution for brand name medicines in all cases where as generic is available but study from India published that 80% of the respondents did not support generic substitution, even in case of prescribed medicines are not available.[36] From our study we can realize that our community pharmacy practitioner has good attitude and awareness toward generic substitution. The current study showed 50.5% of the participants claimed that community pharmacists should be allowed to perform generic substitutions without consulting prescribing physicians which is lower than the report of Turkey that revealed 55% of the respondent’s acclaim that Pharmacist may substitute a generic on his own.[37]
Pharmacy professionals with experience of from 2 to 5 years (AOR=25.620 [1.954-335.896]) and those with more than 5 years (AOR=106.543 [2.375-4779.542) were more likely to have positive attitude toward generic medicines compared to those with work experience of up to 2 years this might be the result of experience, if the pharmacists had more experience in their professional sector they could have better view on generic product.
From findings of the current study 48.3% of respondents claimed that lack of belief in generic medicines is one of the factor during dispensing, this finding is much higher when we compare it with the findings reported from Ireland that depicted 98% of pharmacists believed that generics were of a similar quality to the originator, and 96% stated that they were as effective as the originator.[38] This difference might be the result of difference in awareness of community pharmacist in Ireland and Ethiopia. As we know Ireland is developed countries and the awareness of the practitioner and implementation of quality health policy would be better. Around three fourth (73.6) of the respondents in the current study consider affordability to the customer is important factors for dispensing generic products. Which is lower report in comparison to findings announced from Australia that showed 91.3% of participants claimed generic medicines are less expensive than brand-name medicines.[33] This might be results from extra availability of generic products which reduce price of the generic products.
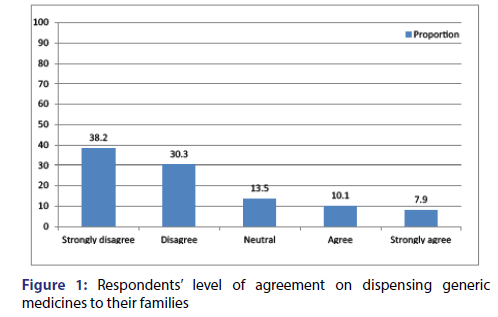
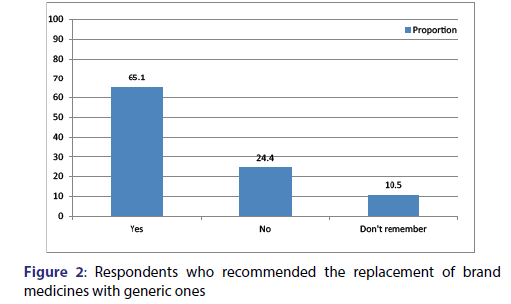
From the current study 55.9% of the respondents [Figures 1 and 2] claimed that locally manufactured generics are equal in their quality compared to the imported generics this result is lower in comparison to findings reported from Kabul that showed 47.5% of the respondents were accepting the concept of locally manufactured generics are equal in their quality compared to the imported generics.[39] From the current study 60.2% of the participants agreed with concept of locally manufactured generics are equal in their safety and efficacy compared to the imported generic this finding is higher in comparison to the report published from Malaysia which depicted 58.4% of the respondents believed locally manufactured generics are equal in their safety and efficacy to the imported generic.[35] Around 63% of the participants in our study claimed that locally manufactured generics are cheaper compared to imported generics and 47.6% of them were considered that imported generics need to pass more stringent approval process compared with locally manufactured ones this result is comparable with findings reported from Malaysia that revealed 47.9% of the respondents agreed with concept of imported generics need to pass more stringent approval process compared with locally manufactured ones [35] this similarity could be result of similarity in economic status of the country that would contribute for similar health structure, function and professional awareness.
Conclusion
From this study it is possible to conclude that more than half of the respondents have had knowledge on the concept of generic medicine and their right to perform generic substitution.Year of experience were depicted as a factor for difference in their knowledge and if the pharmacists had more experience in their professional sector they could have better view on generic product. Nearly half of the participants of this study claimed that lack of belief in generic medicines is one of the factors during dispensing.
Acknowledgements
Deepest gratitude goes to the study participants and for the owner of medicine retail outlets in Mekelle city for their permission to conduct this study.
REFERENCES
- Generic Drugs. http://www.who.int/trade/glossary/story034/en/index.html.
- Dunne S. A review of the differences and similarities between generic drugs and their originator counterparts, including economic benefits associated with usage of generic medicines, using Ireland as a case study. BMC Pharmacol Toxicol 2013;141:1.
- Manzoli L, Flacco ME, Boccia S, D’Andrea E, Panic N, Marzuillo C, et al. Generic versus brand-name drugs used in cardiovascular diseases. Eur J Epidemiol 2016;31:351-68.
- Auta A, Bala ET, Shalkur D. Generic medicine substitution: a cross-sectional survey of the perception of pharmacists in north-central, Nigeria. Med Princ Pract 2013;23:53-8.
- Jamshed S, Hassali M, Ibrahim M, Shafie A, Babar Z. Knowledge, perception and attitude of community pharmacists towards generic medicines in Karachi, Pakistan: a qualitative insight. Trop J Pharm Res 2010;9.
- Homedes N, Ugalde A. Multisource drug policies in Latin America: survey of 10 countries. Bulletin of the World Health Organization, 2005;83:64-70.
- Chong CP, Hassali MA, Bahari MB, Shafie AA. Evaluating community pharmacists’ perceptions of future generic substitution policy implementation: a national survey from Malaysia. Health Policy 2010;94:68-75.
- Questions and answers on generic medicines. 2012.
- Ess SM, Schneeweiss S, Szucs TD. European healthcare policies for controlling drug expenditure. Pharmacoeconomics 2003;21:89-103.
- Al-Gedadi NA, Hassali MA. Pharmacists' views on generic medicines: a review of the literature. J Generic Med 2008;5:209-18.
- Lofgren H. Generic drugs: international trends and policy developments in Australia. Aust Health Rev 2004;27:39-48.
- Ioannides-Demos LL, Ibrahim JE, McNeil JJ. Reference-based pricing schemes. Pharmacoeconomics 2002;20:577-91.
- King DR, Kanavos P. Encouraging the use of generic medicines: implications for transition economies. Croat Med J 2002;43:462-69.
- McGavock H. Strategies to improve the cost effectiveness of general practitioner prescribing. Pharmacoeconomics, 1997;12:307-11.
- Johnston A, Asmar R, Dahlöf B, Hill K, Jones DA, Jordan J, et al. Generic and therapeutic substitution: a viewpoint on achieving best practice in Europe. Br J Clin Pharmacol 2011; 72: 727-30.
- Jacobzone, S., Pharmaceutical policies in OECD countries. 2000.
- Garattini L, Tediosi F. A comparative analysis of generics markets in five European countries. Health Policy 2000;51:149-62.
- Danzon PM, Chao LW. Cross-national price differences for pharmaceuticals: how large, and why?. J Health Econ 2000;19:159-95.
- Danzon PM, Chao LW. Does Regulation Drive Out Competition in Pharmaceutical Markets?*. J Law Econ 2000;43:311-58.
- Standard Treatment Guidelines for General Hospital. 2014, Food, Medicine and Healthcare Administration and Control Authority of Ethiopia: Addis Ababa, Ethiopia.
- National Drug Policy of the Transitional Government of Ethiopia. 1993, The Transitional Government of Ethiopia: Addis Ababa, Ethiopia.
- Birkett DJ. Generics-equal or not?. Aust Prescr 2003;26.
- Lamy PP, Generic equivalents: issues and concerns. Br J Clin Pharmacol 1986;26:309-316.
- Grover P Evaluating pharmacists’ views, knowledge, and perception regarding generic medicines in New Zealand. Res Social Adm Pharm 2011;7:294-305.
- Meredith P. Bioequivalence and other unresolved issues in generic drug substitution. Clin Ther 2003;25:2875-2890.
- Goodwin N. Managing people with long-term conditions. An Inquiry into the Quality of General Practice in England. The King’s Fund, 2010.
- Adelman C, Norris J. Generic medicines are not substandard medicines. The Lancet 2002;359:1351-52.
- Awaisu A. Evaluating community pharmacists' perceptions and practices on generic medicines: A pilot study from Peninsular Malaysia. Journal of Generic Medicines: The Business Journal for the Generic Medicines Sector 2008;5:315-330.
- Awaisu A. Knowledge, attitudes, and practices of community pharmacists on generic medicines in Qatar. Int J Clin Pharm 2014;36:394-404.
- Gebrekirstos H, Kidanemariam A, Gebrezgiabher G. assessment of knowledge, attitude and practice of pharmacists and physicians toward generic medicine use in private clinics and dispensary of mekelle city, Northern Ethiopia.
- Grover P, Stewart J, Hogg M, Short L, Seo HG, Rew A. Evaluating pharmacists’ views, knowledge, and perception regarding generic medicines in New Zealand. Res Social Adm Pharm 2011;7:294-305.
- Chong CP, Hassali MA, Bahari MB, Shafie AA. Exploring community pharmacists’ views on generic medicines: a nationwide study from Malaysia. Int J Clin Pharm 2011;33:124-31.
- Hassali MA, Kong D, Stewart K. A comparison between senior medical students' and pharmacy pre‐registrants' knowledge and perceptions of generic medicines. Med Educ 2007;41:703-10.
- Allenet B, Barry H. Opinion and behaviour of pharmacists towards the substitution of branded drugs by generic drugs: survey of 1,000 French community pharmacists. Pharm World Sci 2003;25:197-202.
- Hassali MA, Shafie AA, Chong CP, Saleem F, Atif M, Chua G, et al. Community pharmacist's perceptions towards the quality of locally manufactured generic medicines: A descriptive study from Malaysia. J Appl Pharm Sci 2012;2:56.
- Basak S. Sathyanarayana D. Exploring knowledge and perceptions of generic medicines among drug retailers and community pharmacists. Indian J Pharm Sci 2012;74:571.
- Toklu HZ, Dülger GA, Hidiroğlu S, Akici A, Yetim A, Gannemoğlu HM, et al. Knowledge and attitudes of the pharmacists, prescribers and patients towards generic drug use in Istanbul–Turkey. Age 2012;25:36-45.
- Othman GQ, Abdulghani MA. Assessment of knowledge and perceptions of generic medicines among pharmacy students in Yemeni universities. Pharm Educ 2015;15.
- Bashaar M, Hassali MA, Saleem F. Community pharmacists’ attitudes toward the quality and price of locally manufactured generic medicines in Kabul, Afghanistan. J Pharm Policy Pract 2015;8:1.