Antidiabetic effect of combined aqueous leaf extract of Vernonia amygdalina and metformin in rats
- *Corresponding Author:
- A.Maxwell Ogochukwu
Department of Clinical Pharmacy and Pharmacy Management, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka 410001, Enugu State, Nigeria.
E-mail: maxolpharmacia@yahoo.com
- C. Okoye Theophine
Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka 410001, Enugu State, Nigeria
E-mail: theokubs@yahoo.com
Date of Received : 20-04-2010
Date of Modefied : 16-05-2010
Date of Accepted : 21-06-2010
Available Online : 15-08-2010
Abstract
This study investigated the antidiabetic activity of the various combina-tions (ratios) of metformin (50 mg/kg) and aqueous extracts of the leaves of Vernonia amygdalina (100 mg/kg). The ratios of Extract to Metformin were 1:1, 2 1, and 2:1 and distilled water (control, p.o.) were given to both normoglycemic and alloxan-induced diabetic Wister albino rats. Blood was withdrawn and tested at 0, 1, 3 and 6 hours. Results showed that the combinations of the extract and metformin caused more re-duction in glycemia compared to any of the agents acting alone in either of the two categories of animals. The ratio of 1:2 caused the most significant (p<0.05) reduction in blood sugar (-66.07%) compared to distilled water (-7.2%). However, the ratio of metformin: extract (2:1) caused a reduction of -62.66% but was found a better combi-nation considering the safety of the drugs. The combination of Vernonia amygdalina with metformin for the management of diabetes should be highly encouraged with a reduction in the dose of metformin and an increase in the dose of the plant extract to guarantee efficacy and safety.
Keywords
Antidiabetic drug, fasting blood sugar, Metformin and Vernonia amygdalina,
Introduction
Diabetes mellitus is a syndrome of impaired carbohydrate, fat and protein metabolism caused by either lack of insulin secretion or decreased sensitivity of the tissues to insulin [1]. It is a metabolic disease with severe socio-economic importance characterized by hyperglycemia and glycosuria due to absolute or relative lack of insulin [2]. On the basis of laboratory findings, World Health Organization (WHO) has defi ned diabetes mellitus as a fasting venous plasma glucose concentration greater than 7.8mmol/l (140mg/dl) or greater than 11.1 mmol/l (200mg/dl) two hours after a carbohydrate meal or two hours after an oral ingestion of the equivalent of 75g glucose, even if the fasting concentration is normal [3]. Recent epidemiological studies indicate that the total number of patients affected by diabetes in 2004 was close to 190 million, a figure likely to have reached 325 million (an increase of more than 70%) by 2005 [4]. Th e figures show that the disease is a global health concern that requires serious effort by all nations towards arresting its scourge. Diabetes is a chronic, incurable disease; therefore treatment is usually directed towards the relief of the depressing symptoms such as hyperglycemia and glycosuria [5]. Conventionally, the management protocol of the disease involves non-pharmacological (diet, exercise and surgery) and/or pharmacological means (insulin and oral hypoglycemic agents). The conventional medical approach of simply using insulin and oral drugs to control diabetes mellitus is not only costly but inadequate, boring and associated with a lot of health risks and complications and these lead to lack of compliance [6,7]. In response to this, the World Health Assembly, in 1989, adopted among its resolutions, the support of national traditional medicine program, drawing attention to herbal medicines as being of great importance to the health of individuals and communities [8]. Plants extracts have been used for a long time as a traditional remedy for diabetes in many parts of the world. Many of them have been investigated. For instance, blueberry leaf extract (mystillin), extract of periwinkle mistletoe [9] were investigated and found to possess antidiabetic properties. In Africa, several plants have been screened based on their usage by the traditional healers for the treatment of diabetes. Such plants include Anacadium occidental, Piclirima netida, Bridelia ferugina, Ginburia alypua, Vernonia amygdalina [10]. Some of them have been found to lower glycemia in both normal and chemical-induced diabetes. Vernonia amygdalina Del. (Astereaceae) popularly known as bitter leaf is widely used for its therapeutic and nutritional purposes. It is a shrub of 2-5 m tall with petiolate leaves of about 6.0mm wide [11]. It is native to the South Eastern part of Nigeria where it is commonly used for preparing soup and has been widely used in folk medicine as anti-malaria, purgative, antiparasitic, treatment of eczema and for maintaining healthy blood glucose levels [3]. Other eff ects reported for the plant include anthelminthic, antitumorigenic [12,13], fever, hiccups, and gastric discomfort [14]. Hence the pharmacodynamic properties of V. amygdalina especially its hypoglycemic activities [15] have received a lot of research attention in recent years. Also V. amygdalina leaf possesses both hypoglycemic and hypolipidemic eff ect and is capable of normalizing other biochemical and hematological abnormalities associated with diabetes mellitus and thus could be prescribed as adjunct to dietary and main therapy for diabetes mellitus [16]. Th e fact that the plant possesses antidiabetic properties and is also commonly used for edible purpose suggests possibility of therapeutic interaction when used together with a conventional antidiabetic agent either consciously or inadvertently. The interaction may be additive, synergistic or antagonistic. There is need, therefore, to carry out a systematic investigation into the eff ect of such combinations. This investigation was aimed at determining the antidiabetic effect of a combination of the extract from the leaves of Vernonia amygdalina with an oral hypoglycemic agent, metformin, which belongs to the class of biguanides as well as the combination ratio that was most appropriate for hyperglycemic control.
Methods
Plant Materials
Th e fresh leaves of V. amygdalina Del. were collected from the natural habitat in Nsukka, Enugu state, Nigeria in the month of November, 2008. Th eir botanical identity was confi rmed by Mr. Ogboso of the Department of Pharmacognosy, University of Nigeria, Nsukka. A voucher specimen of the plant has been deposited in the University of Nigeria, Nsukka.
Preparation of Extract
Th e leaves were sorted out to obtain only the fresh leaves and washed with distilled water without squeezing to remove debris and dust particles. Th ey were shade dried for three days and dried leaves pulverized with a manual blender. A portion (200 g) of the powdered leaves was cold macerated with distilled water for 24 hours and fi ltered to obtain the V. amygdalina aqueous extract (VAE) which was concentrated in a freeze dryer and stored for screening tests. Th e appropriate concentrations of the VAE (43g), referred to as the extract, was made in distilled water for the experiments.
Chemicals
Metformin (Merk) was purchased from a registered Pharmacy shop, Phymex Pharmacy Ltd., Minna, Nigeria. Th e tablets were crushed into powder and appropriate concentrations of it made in distilled water preparatory to administration to the experimental animals. Another material used was alloxan monohydrate (Sigma, St Louis MO, USA) for induction of diabetes in the rats.
Animals
Adult (aged 3-4 months) Wister albino rats weighing 65-200g of either sex were purchased from the disease-free stock of the animal house of the Faculty of Veterinary Medicine, University of Nigeria, Nsukka and used for this study. They were maintained in normal and standard laboratory conditions of temperature (28 ± 2°C) and relative humidity (46 ± 6%) with 12-hour lightdark cycle and adequate ventilation. The animals were fed with commercial diet (Vital Feed Nig. Ltd.) and water, ad libitium. Food was withheld 12 hours before the experiments, but there was free access to water. Permission for the use of animals and animal protocols was obtained from the Animal Ethics Committee of the University of Nigeria, Nsukka, prior to experimentation.
Animal categorization
The animals were allowed 14-day acclimatization period, after which they were randomly divided into two broad categories: non diabetic (normoglycemic) and diabetic (hyperglycemic) rats. For the former category, the blood glucose level was confi rmed using glucometer (One-Touch) to determine the sugar level by withdrawing blood from the tail end and testing. Those with glucose level of 19-39 mg/dL were confi rmed to be in the normoglycemic category and used for the study.
Induction of Diabetes
Diabetes was induced on the latter category by intraperitoneal injection of 150mg/kg body weight of alloxan monohydrate freshly prepared with distilled water as the vehicle. Diabetes was confi rmed three days later in alloxan- induced animals showing Random Blood Glucose (RBG) level ≥ 200 mg/dL by using glucometer to monitor the blood sample from the tail vein.
Animal grouping and experimental design
The normoglycemic category: Twenty four rats included in this category were divided into six groups each consisting of four animals. The various substances were administered as follows:
Group A: 100 mg/kg VAE
Group B: 50 mg/kg metformin,
Group C: VAE + metform (1:1)
Group D: VAE + metformin (2:1)
Group E: VAE+ metformin (1:2)
Group F: distilled water (control).
The hyperglycemic category: Twenty four rats included in this category were divided into six groups consisting of four animals each. The substances were administered similar to the normoglycemic category as follows:
Group G: 80 mg/kg VAE
Group H: 40 mg/kg metformin,
Group I: VAE+ metform (1:1)
Group J: VAE+ metformin (2:1)
Group K: VAE + metformin (1:2)
Group L: distilled water (control).
The administration of both the extract and drug was done by oral route. Blood sample was withdrawn from the tail vein with the aid of a capillary tube and tested using the glucometer. It was withdrawn just before oral administration of substances (0 hour) and at 1, 3 and 6 hours in each case. The percentage of glycemia reduction was calculated at the 6th hour during fasting blood sugar (FBS) monitoring using the formula:

where Go and Gx were the values of 0-hr and 6-hr FBS respectively (17).
Statistical analysis
Th e results are presented as mean ± SEM. Statistical diff erences between means were determined by the student’s t-test and p values less than 0.05 were considered signifi cant.
Results
Antidiabetic effect of the test substances on normoglycemic and hyperglycemic rats
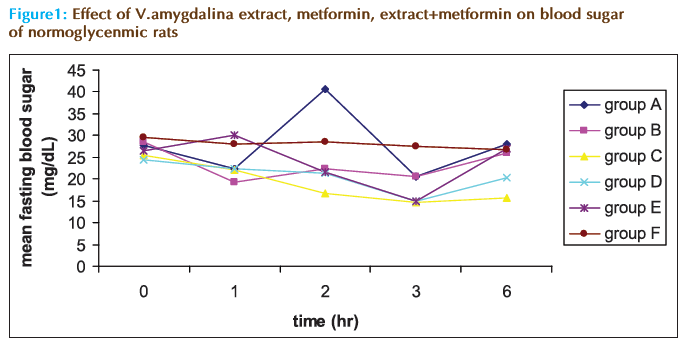
Figure 1 shows the eff ect of VAE, metformin and their various combinations in the mean fasting blood sugar of the non-diabetic rats in comparison with distilled water. It can be seen that while distilled water maintained the sugar level to almost the starting value (29.5 mg/dL) throughout the duration of the monitoring period, the VAE showed signifi cant fl uctuation around the starting value (FBS of 27.75 mg/dL). Metformin alone and the extract–metformin combinations caused a steady and signifi cant reduction in the glycemia in the first five hours. However, after this period they all showed slight increment in the blood sugar level of the animals causing an insignifi cant overall change in glycemia at the 6th hour, as shown in Table 1. From the table, only the combination of extract and metformin in 1:1 (group C) and 2:1 (group D) exhibited signifi cant (p<0.05) reduction in FBS with that of group C being higher (-38%) as against distilled water (-9.32%), metformin alone (-8.77%) and extract alone (+0.9%).
| Treatment | Macrovascular complications | |
|---|---|---|
| Nondiabetic Rats | Diabetic Rats | |
| V. amygdalina (group A and G) | + 0.90 | - 56.69 |
| Metformin (group B and H) | - 8.77 | - 55.66 |
| V.amygdalina + metformin (1:1) (group C and I) | - 38.24 | - 58.41 |
| V. amygdalina +metformin (2:1) (group D and J) | - 17.35 | - 62.66 |
| V. amygdalina+metformin (1:2) (group E and K) | + 1.89 | - 66.07 |
| Distilled H2O (negative control, group F and L) | - 9.32 | - 7.20 |
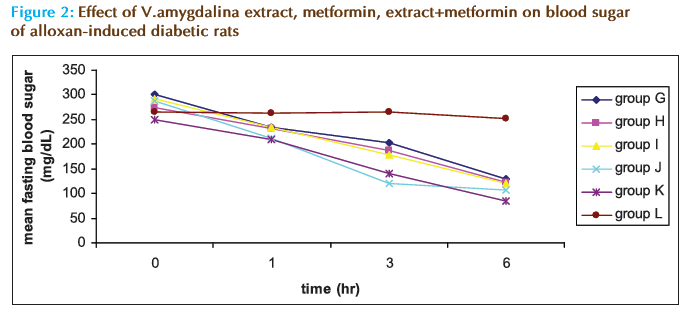
Table 1: The Effect of V. amygdalina, metformin and V. amygdalina with metformin on percentage change of blood glucose in rats
In figure 2 which shows the eff ect of the various substances on the FBS of the diabetic rats, it can be noticed that all the test substances (groups G-K) caused steady and signifi cant hypoglycemia during the monitoring period. Distill water (group L) did not cause signifi cant changes in the glycemia of the animals. Th e overall percent changes present this point clearer (Table 1). It shows that the reduction in the FBS caused by the extract and metformin in any combination is better than that caused by any of them separately though the combination of 1:2 (group J) caused the highest reduction in FBS (-66.07%) compare to distilled water (-7.2%), metformin alone (-55.66%) and extract alone (-55.69%).
Discussion
The effect of the VAE on rats’ blood glucose has implications on its use for nutritional and therapeutic purposes. The observation that the extract did not signifi cantly cause a change in the glucose level of the normoglycemic rats after six hours implies that the extract is safe and does not cause hypoglycemia in a normal subject taking it either as food or for other medical purposes. Apart from its safety in normoglycemic individuals, the extract has a high therapeutic index as the acute toxicity test in rats gave an LD50 of about 1265 mg/kg [8]. In addition, the efficacy of the extract on hyperglycemic rats corroborates the result of other researchers, who had systematically demonstrated that the extract from the plant possesses antidiabetic properties [15,18]. The activity of the extract is attributable to the phytoconstituents. Compounds such as steroid, glycoside and sesquiterpene lactones like vernodalin have been isolated from the plant [19]. Nutritionally, it has been demonstrated that the plant contains moisture and fiber which contribute less sugar to the blood sugar content [6] as plants rich in non starch polysaccharides were reported to reduce postprandial blood glucose concentrations in humans [20]. Other fi ndings have shown that fiber rich food does not raise blood glucose [21,22] rather it enhances insulin sensitivity and may have a role in the prevention and management of type 2 diabetes [23].
The hypoglycemic effect of the combined agents suggests that their antidiabetic activities are addictive and this could mean that the extract and metformin are acting through the same mechanism. According to Shlafer [24], the current proposed mechanism of action of metformin include: direct stimulation of glycolysis in tissues with increase glucose removal from blood; reduced hepatic and renal gluconeogenesis; slowing of glucose absorption from the gastrointestinal tract with increased glucose to lactate conversion by enterocytes; and reduction of plasma glucagon levels. Also its ability to lower glucose levels does not depend on the active pancreatic β-cells [25] unlike other oral agents such as the sulphonylureas. Th is may explain why metformin alone and in combination signifi cantly reduced the blood sugar in the alloxan-induced diabetic rats as alloxan is a known β-cytotoxic agent [26]. The observation that the VAE alone reduced the glycemia signifi cantly in the alloxan treated rats lends credence that it has peripheral action similar to metformin. Other workers in this area have also noted that VAE could have a direct insulin-like effect on glucose metabolism [27,28].
Furthermore, this observed additive effect has a great clinical implication. There is need for collaboration between the traditional medicine and the orthodox medical practitioners in the management of diabetes mellitus. A diabetic patient can be placed on a reduced dose of metformin (which also implies lower adverse eff ect) while being encouraged to consume more of bitter leaf V.amygdalina either in soup, as commonly used by the populace, or in form of extract. Therefore, V. amydalina can play a significant role in the management of either insulin dependent or non insulin dependent diabetes. This is obvious as it is commonly used as diet, which is known to play a central role in diabetic management [29].
The issue of drug-drug interaction should be seriously considered while a patient is combining a potent antidiabetic agent and herb remedies. Adibe M.O et al [30] remarked that cautions should be observed by patients with chronic diseases while using Aloe vera as this may bring about grievous drug- drug interactions and fatal hypersensitivity. The consequences are not different in a diabetic patient combining a potent antidiabetic agent and herbs as this might cause severe hypoglycemia which could lead to coma and ultimately to death.
In a particular patient, medical practitioners should be aware of potential interactions and liaise with appropriate authority or drug advisory department to avoid adverse events which the patient might encounter in course of combining these drugs. Physicians need to specifically ask patients about traditional medicine use and document this appropriately and this should be discussed with the patient in an open non-judgemental manner. Implementation of pharmaceutical care in community pharmacies could help to alleviate this problem. Community pharmacists can play and active role in the provision of advisory and educational services to these patients [31].
Conclusion
Th is investigation has demonstrated that the use of a combination of the extract from the leaves of V. amygdalina and metformin with a greater part of the extract and reduced dose of metformin is quite effi cacious, additive and safe for the management of diabetes mellitus. Th e need for collaboration between the orthodox and herbal practitioners is advocated and increase intake of the leaves of V. amygdalina by both normal and diabetic patients is encouraged.
References
- Guyton AC. Textbook of Medical Physiology. W.B. Sanders Co, London. 1999: 915- 928.
- Aguwa CN. Therapeutic Basis for Clinical Pharmacy in the Tropics. Third edn. SNAAP Press Ltd, Enugu. 2004: 1-230.
- Nwanjo HU, Nwokoro EA. Antidiabetic and biochemical effects of aqueous extract of Vernonia amygdalina leaf in normoglycaemic and diabetic rats. J Innov Life Sci. 2004; 7: 6 - 10.
- Lefebvre P. Endocrine diseases and diabetes mellitus. Diabetes. 2005; 22: 25-35.
- Milton JB. Diabetes: The new approach. Grosset and Duniap Publishers, New York. 1976: 15-17.
- Okolie UV, Okeke CE, Oli JM, et al. Hypoglycemic indices of Vernonia amygdalina on postprandial blood glucose concentration of healthy humans. Afr J Biotech. 2008; 7 (24): 4581-4585.
- Adeneye AA and Agbaje EO. Pharmacological Evaluation of Oral Hypoglycemic and Antidiabetic Effects of Fresh Leaves of Ethanol Extract of Morinda lucida Benth in Normal and Alloxan- Induced Diabetic Rats. Afr. J. Biomed. 2008; 11 (1): 65-71.
- Nwanjo HU. Effi cacy of aqueous leaf extract of Vernonia amygdalina on plasma lipoprotein and oxidative status in diabetic rat models. Nig J Physiol Sci. 2005; 20 (1-2): 39-42.
- Kupchan SM. Drug discovery science and development in a changing society. American Chemical Society, Washington D.C. 1971: 6-8.
- Ogbuokiri, JE and Ekpechi DL. Anti-hyperglycaemic activity of Vernonia amygdalina on healthy adult volunteers. Clin Pharm Herbal Med. 1989; 5(3): 18-21.
- Ojiako OA and Nwanjo HU. Is Vernonia amygdalina hepatotoxic or hepatoprotective Response from biochemical and toxicity studies in rats. Afr J Biotech. 2006; 5(18): 1648-1651.
- Abosi OA and Raseroka BH. In vivo antimalarial activity of Vernonia amygdalina. Brit J Biomed Sci. 2003; 60(2): 89-91.
- Izevbigie EB, Bryant JL and Walker L. A novel natural inhibitor of extracellular signal-regulated kinases and human breast cancer cell growth. Exp Biol Med. 2004; 229 (2): 163-169.
- Hamowia AM and Saffaf AM. Pharmacological studies on Vernonia amygdalina (Del) and Tithonia diversifolia (Gray). Vet Med J Giza. 1994; 2: 91-97.
- Akah PA and Okafor CI. Hypoglycaemic effect of Vernonia amygdalina (Del) in experimental rabbits. Plant Med Res. 1992; 1: 6-10.
- Akah PA, Alemji JA, Salawu OA, et al. Effects of Vernonia amygdalina on Biochemical and Haematological Parameters in Diabetic Rats. Asian J Med Sci. 2009; 1(3): 108-113.
- Fuentes O, Arancibis A and Alarcon H. Hypoglycemic activity of Bauhinia candican in diabetic induced rabbits. Fitoterapis. 2004; 6:527-532.
- Sabu M, and Kuttan R. Antidiabetic activity of medicinal plants and its relationship with their antioxidant property. J Ethnopharmacol. 1982;81: 155-160.
- Kupchan SM, Richard JH, Aziz K, et al. Active constituents of plants. J Org Chem. 1969; 34 (6): 3903-3907.
- Manach C, Scalbert A, Morand C, et al. Polyphenols: food sources and bioavailability. Am J Clin Nutr. 2004; 79: 727- 747.
- Ylonen K, Saloranta C, Kronberg C, et al. Associations of Dietary Fiber with Glucose Metabolism in Non diabetic relatives of subjects with Type 2 Diabetes. Diabetes care. 2003; 26: 1979-1985.
- Oli, J, Ikeakor I, Onwuamaeze I. Blood glucose responses to common Nigerian foods. Trop Geogr Med. 1982; 34: 317-322.
- Henry S. Fiber: An All Natural “Medicine” for Type 2 Diabetes Consumer Health Interactive, 2004. Available at: http://www.ahealthyme. com
- Shlafter M, Marieb NE. The Nurse, Pharmacology and Drug therapy. Addison-Wesley, California. 1989: 905- 930.
- Katzung BG. Basic and Clinical Pharmacology. Mc Grawhill, London. 2001: 711-778.
- Rerup CC. Drugs Producing Diabetes through Damage to the Insulin Secreting Cells. Pharmacological Review. 1970.
- Taiwo IA, Odeigah PGC and Ogunkanmi LA. The Glycaemic Effects of Vernonia amygdalina and V. tenoreana with Tolbutamide in Rats and the Implications for the Treatment of Diabetes Mellitus. J Sci Res Dev. 2009;11: 122 – 130.
- Sepici A, Gurbuz I, Cevik C, et al. Hypoglycemic effects of myrtle oil in normal and alloxan – diabetic rabbits. J Ethnopharmacol. 2004; 93: 311- 318.
- Mann JI. Diet and Diabetes. Diabetologia. 1980; 18: 89-95.
- Adibe Maxwell O, Ukwe Chinwe V and Ekwunife Obinna I. Evaluation of therapeutic uses of Aloe barbadensis miller (aloe vera) plant among staff and students in a Nigerian university. Int. J. Ph. Sci. May-August 2009; 1(1):59-70. Available online from: www.ijps.info.
- Adibe MO. Prevalence of concurrent use of herbal and synthetic medicines among outpatients in a mission hospital in Nigeria. Int. J. Drug Dev & Res. Sep-Dec 2009; 1(1): 60-66. Available online from: www.ijddr.com.